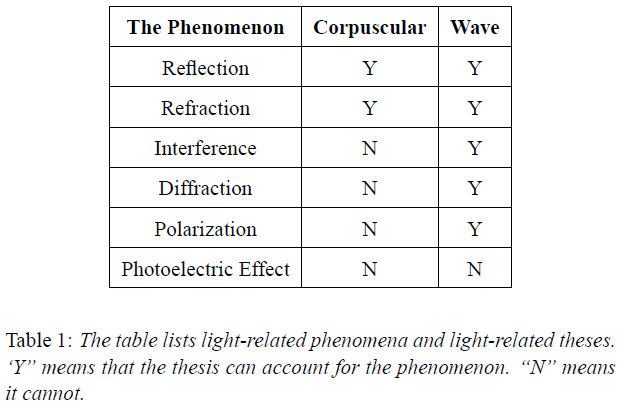
Light (electromagnetic radiation) is a disturbance in space. Light spreads uniformly in space in all directions as it moves away from its source. The intensity of light decreases with distance, but the frequency remains the same. Frequency is the only discrete aspect of light. Each cycle of light represents an energy equal to the Planck’s constant ‘h’.
In 1905, Einstein wrote in his paper “On a heuristic point of view concerning production and transformation of light”:
“It seems to me that the observations associated with blackbody radiation, fluorescence, the production of cathode ray by ultraviolet light, and other related phenomena connected with the emission and transformation of light are more readily understood if one assumes that the energy of light is discontinuously distributed in space. In accordance with the assumption to be considered here, the energy of light ray spreading out from a point source is not continuously distributed over an increasing space but consists of a finite number of energy quanta which are localized at points in space, which move without dividing, and which can only be produced and absorbed as complete units.”
Why did Einstein assume light to be discontinuously distributed in space even when this idea was contradictory to the known wave nature of light?
.
The key reason was that Einstein wanted to explain how light of low intensity could still eject electrons instantaneously when shined over some metals. The energy of the electron followed the equation,
V0.e = hf – φ0
Where
V0.e represents maximum kinetic energy of electron
hf represents energy supplied by the photon of light
φ0 represents energy consumed in ejecting an electron
This relationship was accurately verified by Robert Millikan in 1914 with great precision.
.
Einstein formulated the idea of light quantum (photon) based on the idea of ‘energy quantum’ suggested by Max Planck five years earlier to explain the blackbody radiation.
When heated, a blackbody radiated light at all frequencies. But the frequency distribution of that radiation could not be explained using the principles of classical physics. Max Planck used the “mathematical idea” of energy quantum to resolve the difficulty.
‘hf’ was the energy quantum suggested by Max Planck that was required to activate an “atomic oscillator” of frequency ‘f’, which then radiated light at that frequency.
.
In photoelectric effect, frequency was involved in ejecting the electron and not the intensity of light. Electron could not be ejected by light below a threshold frequency. Energy of the ejected electron then increased only with increasing frequency. An increasing intensity of light did not eject electrons below the threshold frequency. Above that frequency, the increasing intensity simply increased the number of electrons ejected.
At the threshold frequency f0, light supplied the energy hf0 to the atomic oscillator to release the electron. This energy had to be supplied as a bundle to activate the oscillator. So, the energy supplied by light had to be bundled in space.
.
Robert Millikan, who didn’t agree with Einstein’s particle theory of light, analyzed the absorption of light in photoelectric emission (see Section 9, Theories of Photo Emission, of Millikan’s paper). He noted that his experiments showed exact proportionality between the field strength and the force acting on the electron, which contradicted any kind of discontinuous structure in space. This leaves the only alternative that the energy supplied by light must bundle up within the metal.
Frequency triggers resonance in an oscillator of the atom. The initial amplitude of incident light determines how many of those resonances build up and discharge an electron. The amplitude has decreased as the wave front has spread. But it builds up very quickly in a resonating oscillator. The electron discharges at a certain amplitude of oscillator. Energy imparted to an oscillator per cycle of incident light is ‘h’. The more oscillators are exposed to light, the more energy per cycle is imparted. The oscillator discharges at energy ‘hf’.
If we assume that all oscillators in an area can pool their energy to one central oscillator, and that there is one oscillator per atom, then ‘f’ atoms shall be pooling their energy to eject an electron instantly.
With this assumption we can calculate the amount of potassium required to instantly generate a photo electron. For potassium the threshold frequency is 5.537 x 1014 Hertz. One mole of potassium is 39 gram, which consists of 6.022 x 1023 atoms. 5.537 x 1014 atoms of potassium would weigh 3.59 x 10-8 gram. This is an extremely small amount.
Thus, when a very weak star light of threshold frequency falls on a very small area on film of potassium, it can easily eject electrons. We may thus assume that photons are generated upon the interaction of light with the photoelectric material, instead of assuming that photon must already exist in space.
The metallic surface must act as an energy lens to the light shining upon it. The energy carried by the wave front of light is then concentrated at the atomic oscillators within the surface as a photon.
We do not really need to postulate a particle theory of light to explain the photoelectric effect. We may simply postulate that the photon is created as part of the energy interaction.
.





