Reference: Disturbance Theory
.
Black body
-
A black body is an idealized physical body that absorbs all incident electromagnetic radiation, regardless of frequency or angle of incidence.
-
A black body in thermal equilibrium (that is, at a constant temperature) emits electromagnetic radiation called black-body radiation.
-
The radiation has a spectrum that is determined by the temperature alone, not by the body’s shape or composition.
-
It is extremely difficult to realize a perfect black body, for which, the absorption of radiation is 100%. Transmission and reflection is zero.
.
Thermodynamic equilibrium
-
In thermodynamic equilibrium all kinds of equilibrium hold at once.
-
It is characterized by no net macroscopic flows of matter or of energy.
-
Any microscopic exchanges are perfectly balanced.
-
The temperature is spatially uniform.
-
Entropy maximizes with equilibrium.
- A thermodynamic system is a macroscopic object, the microscopic details of which are not explicitly considered in its thermodynamic description.
- It excludes the kinetic energy of motion of the system as a whole and the potential energy of the system as a whole due to external force fields.
- The Boltzmann constant (kB or k), which is named after Ludwig Boltzmann, is a physical constant relating the average kinetic energy of particles in a gas with the temperature of the gas. It is the gas constant R divided by the Avogadro constant NA.
.
Equipartition theorem
-
It relates the temperature of a system to its average energies in thermal equilibrium.
-
It assumes that energy is shared equally among all of its various modes. For example, the average kinetic energy per degree of freedom in translational motion of a molecule should equal that in rotational motion.
-
It gives the average values of individual components of the energy, such as, the kinetic energy of a particular particle, or the potential energy of a single spring. For example, it predicts that every atom in a monatomic ideal gas has an average kinetic energy of (3/2) kBT in thermal equilibrium.
-
When the thermal energy kBT is smaller than the quantum energy spacing in a particular degree of freedom (such as at lower temperatures), the average energy and heat capacity of this degree of freedom are less than the values predicted by equipartition.
-
Such decreases in heat capacity were among the first signs to physicists of the 19th century that classical physics was incorrect and that a new, more subtle, scientific model was required.
-
Along with other evidence, equipartition’s failure to model black-body radiation—also known as the ultraviolet catastrophe—led Max Planck to suggest that energy in the oscillators in an object, which emit light, were quantized, a revolutionary hypothesis that spurred the development of quantum mechanics and quantum field theory.
.
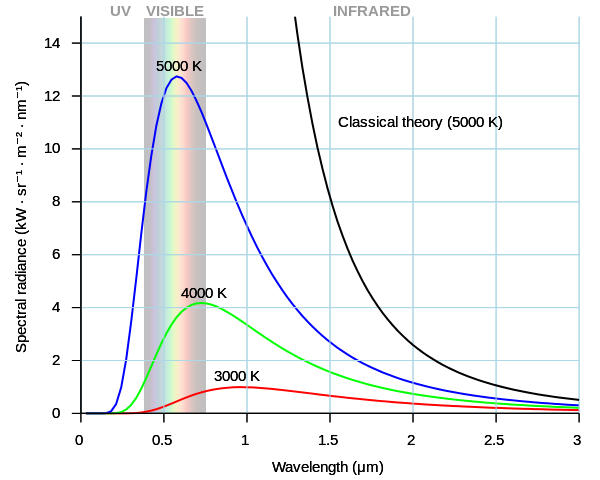
Rayleigh–Jeans law
-
The Rayleigh–Jeans law revealed an important error in physics theory of the time.
-
The law predicted an energy output that diverges towards infinity as wavelength approaches zero (as frequency tends to infinity).
-
Measurements of the spectral emission of actual black bodies revealed that the emission agreed with the Rayleigh–Jeans law at low frequencies but diverged at high frequencies; reaching a maximum and then falling with frequency, so the total energy emitted is finite.
.
Ultraviolet catastrophe
-
The ultraviolet catastrophe was the prediction of classical physics that an ideal black body at thermal equilibrium will emit more energy as the frequency increases.
-
A blackbody would release an infinite amount of energy, contradicting the principles of conservation of energy.
-
The ultraviolet catastrophe results from the equipartition theorem of classical statistical mechanics which states that all harmonic oscillator modes (degrees of freedom) of a system at equilibrium have an average energy of (1/2)kT. It assumes that vibrating modes can increase infinitely.
.
Black-body radiation
-
Black-body radiation is the thermal electromagnetic radiation within or surrounding a body.
-
It has a specific spectrum and intensity that depends only on the body’s temperature.
-
As its temperature increases the peak of the spectrum shifts from infra-red toward higher frequencies of visible light.
-
Black-body radiation has a characteristic, continuous frequency spectrum.
-
If each Fourier mode of the equilibrium radiation in an otherwise empty cavity with perfectly reflective walls is considered as a degree of freedom capable of exchanging energy, then, according to the equipartition theorem of classical physics, there would be an equal amount of energy in each mode.
-
Since there are an infinite number of modes this implies infinite heat capacity (infinite energy at any non-zero temperature), as well as an unphysical spectrum of emitted radiation that grows without bound with increasing frequency, a problem known as the ultraviolet catastrophe.
-
Instead, in quantum theory the occupation numbers of the modes are quantized, cutting off the spectrum at high frequency in agreement with experimental observation and resolving the catastrophe. The study of the laws of black bodies and the failure of classical physics to describe them helped establish the foundations of quantum mechanics.
Explanation
-
The radiation from matter represents a conversion of a body’s thermal energy into radiative energy. At thermal equilibrium, matter emits and absorbs radiative substance. The radiative substance has a characteristic frequency distribution that depends on the temperature only.
-
At thermodynamic equilibrium the amount of every wavelength in every direction of radiative energy emitted by a body at temperature T is equal to the corresponding amount that the body absorbs because it is surrounded by light at temperature T.
-
The black-body curve is characteristic of thermal light, which depends only on the temperature of the body. The principle of strict equality of emission and absorption is always upheld in a condition of thermodynamic equilibrium.
-
By making changes to Wien’s radiation law consistent with thermodynamics and radiation, Planck found a mathematical expression fitting the experimental data satisfactorily. Planck had to assume that the energy of the oscillators in the cavity was quantized, i.e., it existed in integer multiples of some quantity.
-
Einstein built on this idea and proposed the quantization of radiative energy itself in 1905 to explain the photoelectric effect.
-
These theoretical advances eventually resulted in the superseding of classical electromagnetism by quantum electrodynamics. These quanta were called photons and the black-body cavity was thought of as containing a gas of photons.
-
In addition, it led to the development of quantum probability distributions, called Fermi–Dirac statistics and Bose–Einstein statistics, each applicable to a different class of particles, fermions and bosons.
Also see: Classical to Quantum Mechanics
.
