Reference: Evolution of Physics
This paper presents Chapter II, section 3 from the book THE EVOLUTION OF PHYSICS by A. EINSTEIN and L. INFELD. The contents are from the original publication of this book by Simon and Schuster, New York (1942).
The paragraphs of the original material (in black) are accompanied by brief comments (in color) based on the present understanding. Feedback on these comments is appreciated.
The heading below is linked to the original materials.
.
The First Serious Difficulty
We are now ready to note the first grave difficulty in the application of our general philosophical point of view. It will be shown later that this difficulty, together with another even more serious, caused a complete breakdown of the belief that all phenomena can be explained mechanically.
The mechanical view is to describe all phenomena by means of attractive and repulsive forces depending only on distance and acting between unchangeable particles. This view has serious flaws.
The tremendous development of electricity as a branch of science and technique began with the discovery of the electric current. Here we find in the history of science one of the very few instances in which accident seemed to play an essential role. The story of the convulsion of a frog’s leg is told in many different ways. Regardless of the truth concerning details, there is no doubt that Galvani’s accidental discovery led Volta at the end of the eighteenth century to the construction of what is known as a voltaic battery. This is no longer of any practical use, but it still furnishes a very simple example of a source of current in school demonstrations and in textbook descriptions.
Galvani’s observation of the convulsion of a frog’s leg led Volta to construct a voltaic battery as a source of electric current. This accidental discovery of the electric current led to the tremendous development of electricity as a branch of science.
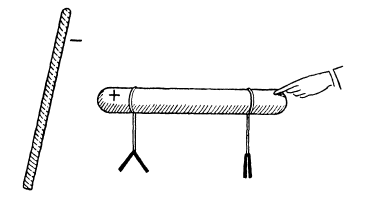
The principle of its construction is simple. There are several glass tumblers, each containing water with a little sulphuric acid. In each glass two metal plates, one copper and the other zinc, are immersed in the solution. The copper plate of one glass is connected to the zinc of the next, so that only the zinc plate of the first and the copper plate of the last glass remain unconnected. We can detect a difference in electric potential between the copper in the first glass and the zinc in the last by means of a fairly sensitive electroscope if the number of the “elements”, that is, glasses with plates, constituting the battery, is sufficiently large.
The voltaic battery, constructed with two different metals in a solution of acid, produces enough electric potential difference between those metals to be detected by an electroscope.
It was only for the purpose of obtaining something easily measurable with apparatus already described that we introduced a battery consisting of several elements. For further discussion, a single element will serve just as well. The potential of the copper turns out to be higher than that of the zinc. “Higher” is used here in the sense in which +2 is greater than -2. If one conductor is connected to the free copper plate and another to the zinc, both will become charged, the first positively and the other negatively. Up to this point nothing particularly new or striking has appeared, and we may try to apply our previous ideas about potential differences. We have seen that a potential difference between two conductors can be quickly nullified by connecting them with a wire, so that there is a flow of electric fluid from one conductor to the other. This process was similar to the equalization of temperatures by heat flow. But does this work in the case of a voltaic battery? Volta wrote in his report that the plates behave like conductors:
. . . feebly charged, which act unceasingly or so that their charge after each discharge re-establishes itself; which, in a word, provides an unlimited charge or imposes a perpetual action or impulsion of the electric fluid.
The electric potential difference was expected to discharge when the two metals were connected, but, instead, there was an unceasing flow of charge.
The astonishing result of his experiment is that the potential difference between the copper and zinc plates does not vanish as in the case of two charged conductors connected by a wire. The difference persists, and according to the fluids theory it must cause a constant flow of electric fluid from the higher potential level (copper plate) to the lower (zinc plate). In an attempt to save the fluid theory, we may assume that some constant force acts to regenerate the potential difference and cause a flow of electric fluid. But the whole phenomenon is astonishing from the standpoint of energy. A noticeable quantity of heat is generated in the wire carrying the current, even enough to melt the wire if it is a thin one. Therefore, heat-energy is created in the wire. But the whole voltaic battery forms an isolated system, since no external energy is being supplied. If we want to save the law of conservation of energy we must find where the transformations take place, and at what expense the heat is created. It is not difficult to realize that complicated chemical processes are taking place in the battery, processes in which the immersed copper and zinc, as well as the liquid itself, take active parts. From the standpoint of energy this is the chain of transformations which are taking place: chemical energy → energy of the flowing electric fluid, i.e., the current → heat. A voltaic battery does not last for ever; the chemical changes associated with the flow of electricity make the battery useless after a time.
From the standpoint of conservation of energy this is the chain of transformations which are taking place: chemical energy → energy of the flowing electric fluid, i.e., the current → heat.
The experiment which actually revealed the great difficulties in applying the mechanical ideas must sound strange to anyone hearing about it for the first time. It was performed by Oersted about a hundred and twenty years ago. He reports:
By these experiments it seems to be shown that the magnetic needle was moved from its position by help of a galvanic apparatus, and that, when the galvanic circuit was closed, but not when open, as certain very celebrated physicists in vain attempted several years ago.

Suppose we have a voltaic battery and a conducting wire. If the wire is connected to the copper plate but not to the zinc, there will exist a potential difference but no current can flow. Let us assume that the wire is bent to form a circle, in the centre of which a magnetic needle is placed, both wire and needle lying in the same plane. Nothing happens so long as the wire does not touch the zinc plate. There are no forces acting, the existing potential difference having no influence whatever on the position of the needle. It seems difficult to understand why the “very celebrated physicists”, as Oersted called them, expected such an influence.
But now let us join the wire to the zinc plate. Immediately a strange thing happens. The magnetic needle turns from its previous position. One of its poles now points to the reader if the page of this book represents the plane of the circle. The effect is that of a force, perpendicular to the plane, acting on the magnetic pole. Faced with the facts of the experiment, we can hardly avoid drawing such a conclusion about the direction of the force acting.
The moment the circular wire is joined to the zinc plate, the magnetic needle turns, as if there is a force acting on the magnetic pole, perpendicular to the plane of the wire.
This experiment is interesting, in the first place, because it shows a relation between two apparently quite different phenomena, magnetism and electric current. There is another aspect even more important. The force between the magnetic pole and the small portions of the wire through which the current flows cannot lie along lines connecting the wire and needle, or the particles of flowing electric fluid and the elementary magnetic dipoles. The force is perpendicular to these lines! For the first time there appears a force quite different from that to which, according to our mechanical point of view, we intended to reduce all actions in the external world. We remember that the forces of gravitation, electrostatics, and magnetism, obeying the laws of Newton and Coulomb, act along the line adjoining the two attracting or repelling bodies.
Here we see a relation between two apparently quite different phenomena, magnetism and electric current. Furthermore, the force is perpendicular to the lines connecting the wire and the needle, unlike the mechanical forces.
This difficulty was stressed even more by an experiment performed with great skill by Rowland nearly sixty years ago. Leaving out technical details, this experiment could be described as follows. Imagine a small charged sphere. Imagine further that this sphere moves very fast in a circle at the centre of which is a magnetic needle. This is, in principle, the same experiment as Oersted’s, the only difference being that instead of an ordinary current we have a mechanically effected motion of the electric charge. Rowland found that the result is indeed similar to that observed when a current flows in a circular wire. The magnet is deflected by a perpendicular force.

Let us now move the charge faster. The force acting on the magnetic pole is, as a result, increased; the deflection from its initial position becomes more distinct. This observation presents another grave complication. Not only does the force fail to lie on the line connecting charge and magnet, but the intensity of the force depends on the velocity of the charge. The whole mechanical point of view was based on the belief that all phenomena can be explained in terms of forces depending only on the distance and not on the velocity. The result of Rowland’s experiment certainly shakes this belief. Yet we may choose to be conservative and seek a solution within the frame of old ideas.
Not only does the force fail to lie on the line connecting charge and magnet, but the intensity of the force depends on the velocity of the charge, and not on the distance as is the case with mechanical forces.
Difficulties of this kind, sudden and unexpected obstacles in the triumphant development of a theory, arise frequently in science. Sometimes a simple generalization of the old ideas seems, at least temporarily, to be a good way out. It would seem sufficient in the present case, for example, to broaden the previous point of view and introduce more general forces between the elementary particles. Very often, however, it is impossible to patch up an old theory, and the difficulties result in its downfall and the rise of a new one. Here it was not only the behaviour of a tiny magnetic needle which broke the apparently well-founded and successful mechanical theories. Another attack, even more vigorous, came from an entirely different angle. But this is another story, and we shall tell it later.
Here we observe a phenomenon that contradicts the mechanical view.
.
Final Comment
The mechanical view describes all phenomena by means of attractive and repulsive forces that depend only on distance and act between unchangeable particles. This view is seriously violated, when we observe that a charge, moving circularly in a plane, generates a force perpendicular to that plane. Furthermore, the intensity of this force depends on the velocity of the charge, and not on any distance.
.