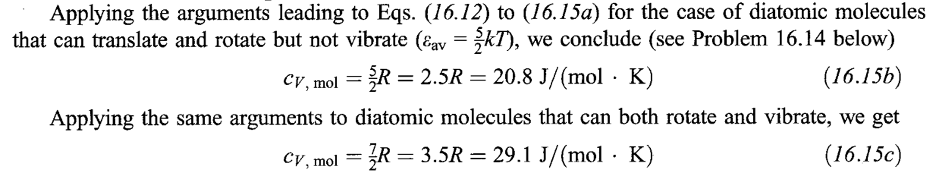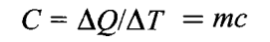Reference: Beginning Physics I
CHAPTER 15: THERMODYNAMICS I: TEMPERATURE & HEAT
.
KEY WORD LIST
Macroscopic Systems, Thermodynamics, Quasistatic Systems, Thermodynamic Variables, Mechanical Equilibrium, Chemical Equilibrium, Thermal Equilibrium, Thermodynamic Equilibrium, Temperature, Zeroth Law of Thermodynamics, Thermometric Property, Celsius Scale, Ice Point, Steam Point, Fahrenheit Scale, Thermometer, Constant Volume Gas Thermometer, Kelvin Temperature Scale, Rankine Temperature Scale, Triple Point, Coefficient of Linear Expansion, Caloric, Thermal Energy, Heat, Internal Energy, Calorie, British Thermal Unit, Specific Heat, Heat Capacity, Calorimetry, Heat of Fusion, Heat of Vaporization, Heat of Sublimation, P-T Diagram, Evaporation
.
GLOSSARY
For details on the following concepts, please consult CHAPTER 15.
MACROSCOPIC SYSTEMS
These are large systems that are characterized by their having myriad atoms and/or molecules. They depend on the myriad random motions and interactions of the component atoms and molecules, rather than their lockstep behavior.
THERMODYNAMICS
Thermodynamics deals with macroscopic systems. More specifically, it deals with the relations between heat and other forms of energy (such as mechanical, electrical, or chemical energy), and, by extension, of the relationships between all forms of energy.
QUASISTATIC SYSTEMS
Quasistatic systems mean that they are in mechanical, chemical, and thermal equilibrium, or that their properties vary so slowly that they can be described at any instant as if in equilibrium.
THERMODYNAMIC VARIABLES
Thermodynamic variables (or macroscopic variables) are physical properties, such as, volume and internal energy, which describe the system as a whole. Most other thermodynamic variables, such as, pressure and temperature can be defined only if the system is quasistatic.
MECHANICAL EQUILIBRIUM
This is understood to mean not only that the system as a whole does not accelerate, but that within the system the different parts are in mechanical equilibrium with each other —no churning of fluids and no pressure imbalances.
CHEMICAL EQUILIBRIUM
A system in mechanical equilibrium may still undergo change through a chemical reaction. The system is in chemical equilibrium when there is no change in chemical composition taking place.
THERMAL EQUILIBRIUM
A system in mechanical and chemical equilibrium may still undergo change in temperature. Two objects in thermal equilibrium with each other are also said to be at the same temperature.
THERMODYNAMIC EQUILIBRIUM
A system that is in mechanical, chemical and thermal equilibrium with its surroundings, as well as internally is said to be in thermodynamic equilibrium. Thermodynamic equilibrium means that there is no change on the macroscopic level.
TEMPERATURE
Temperature is a numerical value that we assign to each thermal equilibrium state of a system as determined by some agreed-upon procedure.
ZEROTH LAW OF THERMODYNAMICS
If two systems A and B are each found to be in thermal equilibrium with a third system C, then when the two systems A and B are brought into contact with each other, they are themselves found to be in thermal equilibrium.
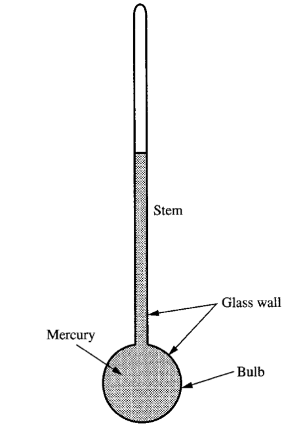
THERMOMETRIC PROPERTY
A thermometric property is a property that varies with the thermal equilibrium states in a well-defined and reproducible way. For example, mercury in a sealed hollow bulb attached to a long, thin hollow glass stem. When mercury expands or contracts with change in the thermal equilibrium state, small changes in its volume are observable from its height in the thin stem.
CELSIUS SCALE
This is the most widely used temperature scale that assigns the number tC = 0°C for the ice point, and tC =100°C for steam point at atmospheric pressure. The distance between these two points is divided into 100 equal marked intervals labeled in 1°C steps.
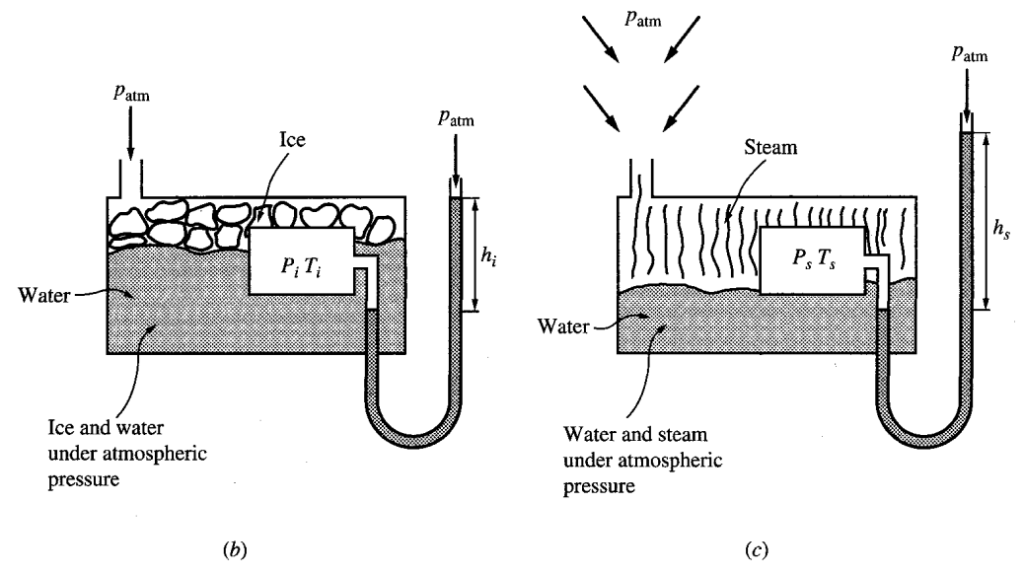
ICE POINT
The point at which ice and water are in thermal equilibrium at atmospheric pressure.
STEAM POINT
The point at which steam, and water are in thermal equilibrium at atmospheric pressure.
FAHRENHEIT SCALE
On this scale the ice point and steam point are defined as tF = 32°F and tF = 212°F respectively, and the distance between these two points is divided into 180 equal marked intervals labeled in 1°F steps.
THERMOMETER
A thermometer is a temperature-calibrated mercury system, which can be used to measure the temperature of any other object. However, the temperature scale shall be dependent on the material being used to define it.
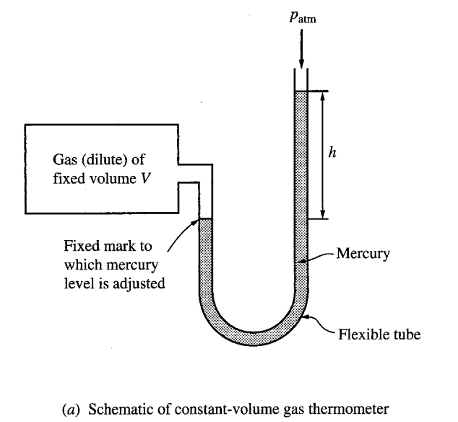
CONSTANT VOLUME GAS THERMOMETER
This thermometer consists of a gas confined to a fixed volume, with an open-tube manometer used to measure the pressure of the gas inside. Constant volume gas thermometer is often considered the “standard” against which other thermometers are calibrated.
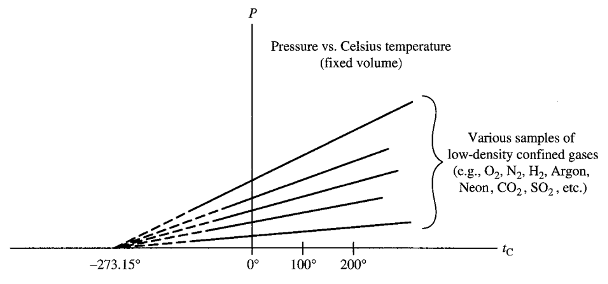
KELVIN TEMPERATURE SCALE
The graphs of pressure vs. temperature of all very low-density gases at fixed volumes are straight lines. When extrapolated these straight lines intersect the temperature axis at the same point: -273.15°C. On the basis of this result, one defines the Kelvin (or absolute) temperature scale. It is same as the Celsius Scale with its zero shifted to “-273.15°C”.
T = tC + 273.15
RANKINE TEMPERATURE SCALE
This is the Kelvin scale using the Fahrenheit degree rather than the Celsius degrees.
TR = tF + 459.67
TRIPLE POINT
The triple point is the temperature, tC = 0.01°C, at which all three phases of water—solid, liquid, and vapor—coexist.
COEFFICIENT OF LINEAR EXPANSION
If we have a rod of length L at a given absolute temperature and we increase the temperature by a small amount ∆T, we find that the length of the rod increases b an amount ∆L that is proportional to the original length L and to the temperature increase ∆T:
∆L = a L∆T
The proportionality constant a is called the coefficient of linear expansion; it depends on the material of which the rod is made.
CALORIC
Early scientists believed that some invisible and weightless substance, which they called caloric, flows from a hotter to cooler object until both objects reach thermal equilibrium.
THERMAL ENERGY
It became clear through the efforts of Joule and others that it is not caloric but thermal energy that is transferred between two macroscopic systems in contact.
HEAT
Heat is the thermal energy transfer from one system to another. Heat is actually the statistical “summing up” of the mechanical work done by the random interactions of the individual atoms and molecules of the two systems.
INTERNAL ENERGY
Related to heat is the internal energy that resides in a system due to the random motion and jiggling of the myriad atoms and molecules making up that system.
CALORIE (CAL)
A calorie is defined as the “amount of heat” (thermal energy in transit) necessary (at atmospheric) to raise the temperature of 1 gram of water 1°C. 1 cal = 4.184 J
BRITISH THERMAL UNIT (BTU)
A Btu is the amount of heat necessary to raise 1 lb of water 1°F. The conversion is 1 Btu = 252 cal.
SPECIFIC HEAT (c)
The specific heat is the characteristic amount of heat that flows into a unit mass of a given substance and raises its temperature by 1°. For solids and liquids, heat is transferred under constant atmospheric pressure.
HEAT CAPACITY (C)
Heat capacity is the total amount of heat needed to produce a degree rise in temperature.
CALORIMETRY
Calorimetry is the experimental measurements of specific heats and other heat constants.
HEAT OF FUSION (Lf)
Heat of fusion is the amount of heat added to melt each unit mass of substance at the melting point (under normal atmospheric pressure).
HEAT OF VAPORIZATION (Lv)
Heat of vaporization is the amount of heat added to vaporize each unit mass of substance at the boiling point (under normal atmospheric pressure).
HEAT OF SUBLIMATION (Ls)
Heat of sublimation is the amount of heat added to sublimate each unit mass of substance at the sublimation point (under normal atmospheric pressure).
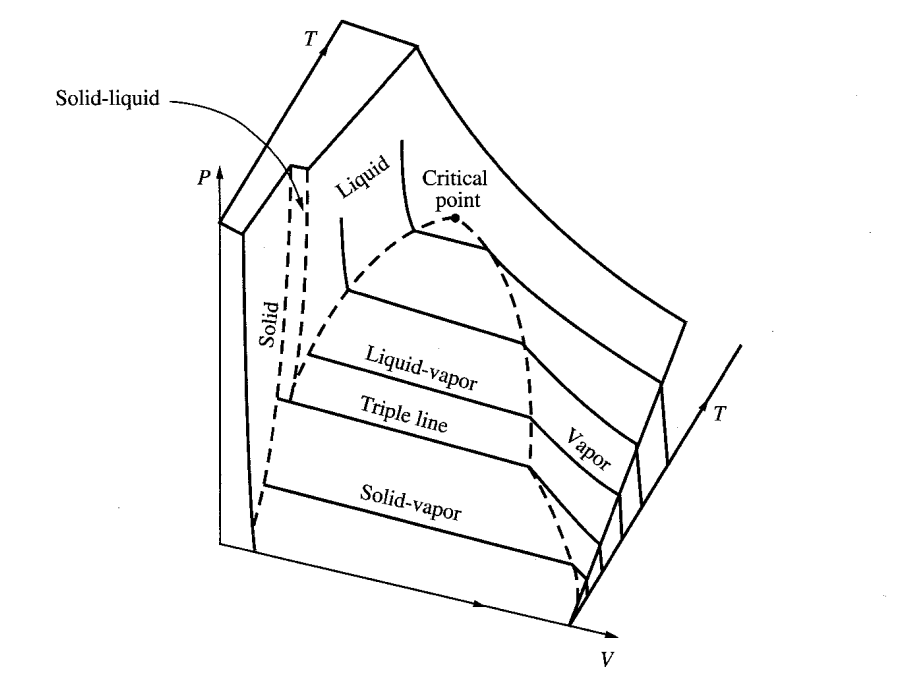
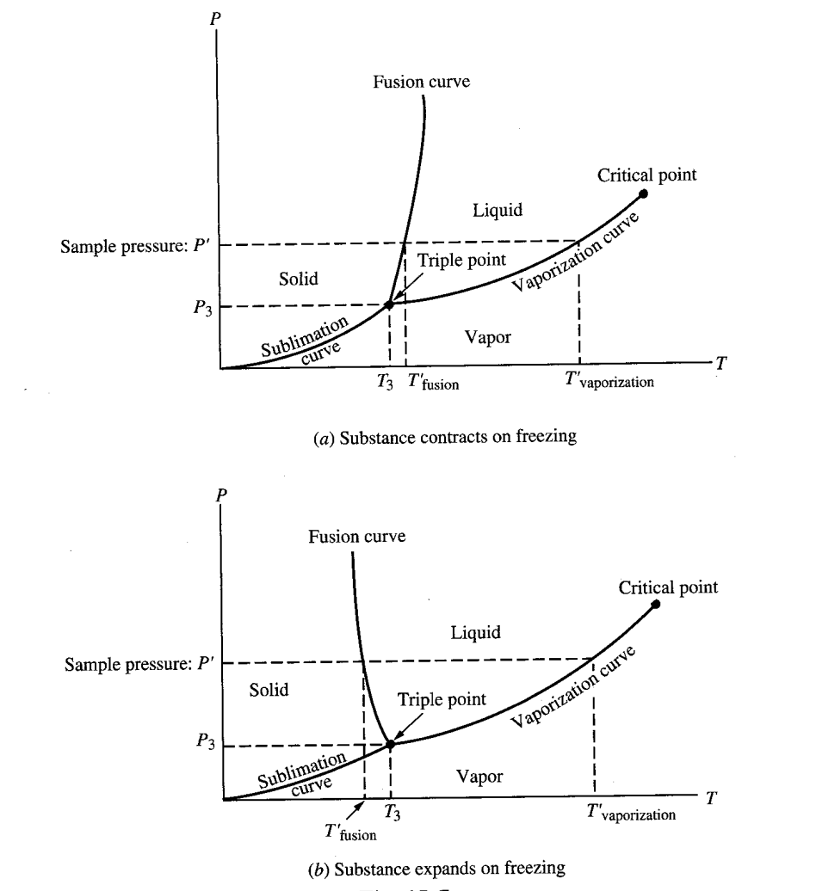
P-T DIAGRAM
The P-T diagram keeps track of phase changes. For a pure substance the diagram will resemble the following.
EVAPORATION
Evaporation takes place at the surface of the liquid in contact with a gas at a given pressure. At temperatures well below the boiling point, molecules from the liquid that are particularly energetic can break free and rise above the liquid to form a vapor. The evaporating molecules take away the thermal energy with them—on average the amount of energy per unit mass is the same order of magnitude as the heat of vaporization for boiling. Thus the evaporation process removes heat from the liquid, cooling it and anything in contact with it.
.