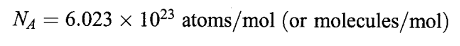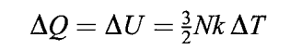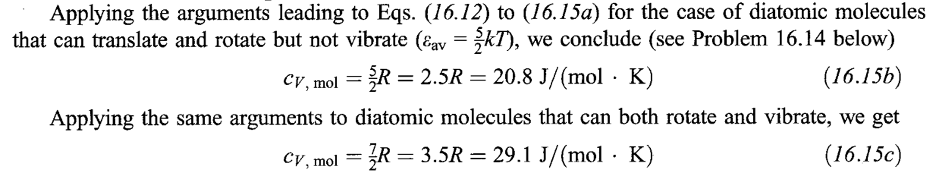Reference: Beginning Physics I
CHAPTER 16: THERMODYNAMICS II: GAS LAWS, THE ATOMIC VIEW, AND STATISTICAL MECHANICS
.
KEY WORD LIST
Molecular Mass, Mole, Avogadro’s Number, Boltzmann Constant, Universal Gas Constant, Ideal Gas Law, Average Kinetic Energy, Mean Square Velocity, Internal Energy, Heat Capacity, Molar Heat Capacity, Equipartition of Energy, Statistical Mechanics, Law of Dulong and Petit
.
GLOSSARY
For details on the following concepts, please consult CHAPTER 16.
MOLECULAR MASS
A mass of any substance whose numerical value in grams is the same as its atomic or molecular mass is called a gram atomic or molecular mass of that substance.
MOLE
One gram atomic or molecular mass of any substance is called a mole of that substance.
AVOGADRO’S NUMBER (NA)
The number of atoms or molecules in a mole of any substance is this a universal constant called Avogadro’s Number.
BOLTZMANN CONSTANT (k)
This is a universal constant:
k = 1.38 x 10-23 J/(particle. K)
UNIVERSAL GAS CONSTANT (R)
R = NAk = 8.31 J / (mol . K)
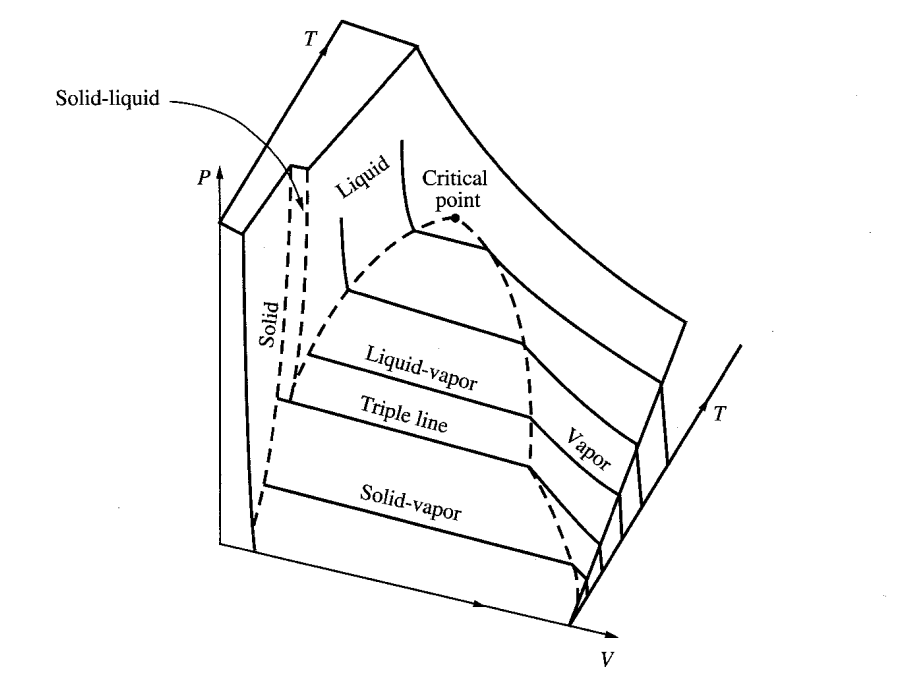
IDEAL GAS LAW
For any confined diluted gas:
PV = nRT
The result from the laws of statistical mechanics is
AVERAGE KINETIC ENERGY
The average translational kinetic energy per molecule in a sample of ideal gas is
This gives a fundamental meaning to the concept of temperature.
MEAN SQUARE VELOCITY
The mean square velocity (v2)av is the average value of the square of the magnitude of velocity of the gas molecules.
At a given temperature the lighter molecules have greater velocities since the average kinetic energy is the same for all gases at a given temperature.
INTERNAL ENERGY (U)
In our infinitesimal “billiard ball” model of a monoatomic gas, the only energy is translational kinetic energy. Therefore, the internal energy is:
If we add some heat to our system, we must have,
HEAT CAPACITY (C)
Heat capacity is the total amount of heat needed to produce a degree rise in temperature. For a constant volume process,
MOLAR HEAT CAPACITY (cv)
The heat capacity per mole for an ideal gas at constant volume:
The heat capacity per mole for an ideal gas at constant pressure:
EQUIPARTITION OF ENERGY
These results, in which each degree of freedom that involves energy (with certain restrictions) contribute the same value (1/2 kT) to the average energy, are called the law of equipartition of energy.
STATISTICAL MECHANICS
Thermodynamics and statistical mechanics thus allowed for the indirect study of the physics of the realm of atoms and molecules, which lead to the realization that Newtonian mechanics does not apply in this realm. This in turn led to the formulation of the new “quantum” mechanics in the early twentieth century.
LAW OF DULONG AND PETIT
A study of the actual values of the molar heat capacities of crystalline solids at constant volume shows that at high temperatures they all have essentially the same molar heat capacity 3R (six degrees of freedom). But all real crystal solids have molar heat capacities that decrease to zero as the Kelvin temperature decreases to zero. This is because the assumption of Newtonian mechanics do not hold.
.