
Reference: Holding: Blank Glossary
- Science, literally means, “knowledge.”
- Philosophy is synthetic interpretation as wisdom,
- But science is analytical description as knowledge.
- Science is defined by the Scientific Method.
- Its purpose is to discover the precise laws of nature.
- .
- .
- Science begins with hypotheses of reasoned conjecture.
- And develops theories demonstrable by experiments.
- But the theories are never absolute, and exploration continues.
- Until solutions to complex life situations are found,
- That help human life survive and evolve further.
- .
- .
- But science has focused on physical phenomena only.
- It has not applied its method to the thought phenomena
- For it does not know how to measure thought.
- But the scientific method expands into the principle of Oneness.
- The criterion of continuity, consistency and harmony measures truth.
- .
- .
- Michael Faraday insisted upon the principle of oneness
- In his essay: On the Conservation of Force.
- But that call was ignored by the scientific community
- Who have yet to accept ‘energy’ as substance.
- And who are unable to see ‘thought’ as substance.
- .
- .
- The thought phenomena underlies the physical phenomena.
- It is now being investigated as the information technology.
- Thought is as much a substance as energy and matter.
- The conservation principle of physics should now be
- The conservation of matter, energy and thought.
- .
- .
.
Key Concepts
.
SECTION 0: GENERAL
ENERGY
Energy, in general, refers purely to the intrinsic motion of substance. Matter has energy. Radiation has anergy. Thought has energy. Both mass (degree of substantiality) and energy (degree of motion) are inherent to substance. They are different concepts and should not be confused with each other as common in physics when talking about radiation. The intrinsic motion gives an object certain intrinsic kinetic energy (KE). KE can be added to an object pushing it such that it gains velocity. The KE of an object can be made to appear zero in an inertial reference frame that is moving at the same velocity as the object. By contrast, the total kinetic energy of a system of objects cannot be reduced to zero by a suitable choice of the inertial reference frame, when the objects are moving at different velocities. But there can be a non-zero minimum. This minimum kinetic energy contributes to the system’s invariant mass, which is independent of the reference frame.
FORCE
Origin: “strong.” Force is an interaction (a push or a pull) that can change an object’s motion or its shape. More formally, it is any action that tends to accelerate a body, deform it, or maintain/alter its state of motion. A force can cause an object to start moving, stop moving, speed up, slow down, change direction, or deform (stretch, compress, twist). In introductory terms, whenever two objects interact—like your hand pushing a door—there is a force involved in that interaction. Force is a vector quantity: it has both magnitude (how strong) and direction (which way it acts). To predict motion, one must add all forces as vectors to get the net (resultant) force acting on the body.
GRAVITY
Gravity is a phenomenon very similar to inertia. Inertia acts to restore the equilibrium between the thickness and motion of a body. Similarly, gravity acts to restore the equilibrium of the distribution of thickness and motion among the bodies of a system.
INVARIANTS
An invariant is a property, quantity, or condition that remains unchanged when a specific transformation, operation, or process is applied to a mathematical object or system.
LOCALITY
The principle of locality states that objects are only directly influenced by their immediate surroundings, requiring physical interactions to propagate through space at or below the speed of light. It rejects instantaneous “action at a distance”. While classical physics is local, quantum entanglement demonstrates non-local correlations.
MECHANICS
Mechanics (from Ancient Greek ‘of machines’) is the area of physics concerned with the relationships between force, matter, and motion among physical objects. Forces applied to objects may result in displacements, which are changes of an object’s position relative to its environment. Mechanics is built on the idea that the motion of material bodies is governed by precise, quantitative relationships between forces, mass, and the resulting motion (or equilibrium) of those bodies. There exist universal laws (classically, Newton’s laws) and conservation principles (energy, momentum, angular momentum) that connect force, mass, and motion in a mathematically exact way, letting us predict how systems will evolve.
MOMENTUM
Momentum in physics is the quantity of motion an object has, defined as the product of its mass and its velocity; it measures how hard it is to stop or change that motion. Momentum points in the same direction as the motion and its magnitude grows with both mass and speed. Momentum extends the idea of inertia (resistance to change of state) to moving bodies. A more massive or faster object has more momentum, so it takes a greater or longer‑acting force to significantly change its motion (for example, stopping a truck vs. a bicycle at the same speed). Force is the rate of change of momentum. In a closed system with no net external force, total momentum remains constant; this is the law of conservation of momentum. Collisions and interactions redistribute momentum among objects, but the vector sum of all momenta before and after the interaction is the same. Momentum is connected to the very structure of the physical law.
WORK
Work is the transfer of energy that happens when a force causes an object to move over some distance in the direction of that force (W= Fd cosθ). There may be force, but if the displacement is zero, or it is perpendicular to the force, then the work done is zero. Work and energy share the same unit because the net work done on a particle equals its change in kinetic energy. Work is positive when energy is added to the object as in accelerating a car forward. Work is negative when energy is removed as in friction slowing a sliding block. Work has magnitude and sign (positive or negative) but no direction in space. In general, work is the mechanical effort required to change a system from one state to another. In thermodynamics, work performed by a system is energy transferred by the system to its surroundings, by a mechanism through which the system can spontaneously exert macroscopic forces on its surroundings, where those forces, and their external effects, can be measured.
.
.
.
PARTICLE
Please see Glossary: Postulate Mechanics.
PRESSURE
Pressure is the force applied perpendicular to the surface of an object per unit area over which that force is distributed.
SPEED
Average speed is defined as the total distance traveled in a given time divided by that time interval. Since distance traveled is always positive, the average speed is always positive. Its units are the same as those of velocity. Average speed is either equal to or greater than the average velocity. Instantaneous speed is always the same as instantaneous velocity.
TIME
Time is a measure of when the object is at a certain position in a coordinate system. Time elapsed is always positive.
VELOCITY
Velocity is the time rate of change of displacement.
WAVE
WAVES are cycles of oscillations that have the characteristics of a continuum.
A wave can be a disturbance traveling in a stationary medium, such as, the waves on the surface of water in a pond.
A wave can also be a rapidly traveling substance with the characteristics of a continuum, such as, light or the rolling waves of the sea.
.
SECTION 3: THERMODYNAMICS
THERMODYNAMICS, HEAT, TEMPERATURE
.
ADIABATIC PROCESS
An adiabatic process occurs without transferring heat or mass between a thermodynamic system and its surroundings. Unlike an isothermal process, an adiabatic process transfers energy to the surroundings only as work. It also conceptually forms the foundation of the theory used to expound the first law of thermodynamics and is therefore a key thermodynamic concept. Q = 0 but ΔT ≠ 0.
CARNOT CYCLE
The Carnot Cycle is a theoretically reversible cycle in which entropy is conserved. The cycle operates between two “heat reservoirs” at temperatures Th and Tc (hot and cold respectively). The reservoirs have such large thermal capacity that their temperatures are practically unaffected by a single cycle.
During the Carnot cycle, an amount of energy ThΔS is extracted from the hot reservoir and a smaller amount of energy TcΔS is deposited in the cold reservoir. The difference in the two energies (Th-Tc)ΔS is equal to the work done by the engine. Thus, heat is converted into work.
ENERGY, INTERNAL
The internal energy keeps account of the gains and losses of energy of the system that are due to changes in its internal state. It is often not necessary to consider all of the system’s intrinsic energies. Internal energy is measured as a difference from a reference zero defined by a standard state. The processes that define the internal energy in the state of interest are transfers of matter, or of energy as heat, or as thermodynamic work. If the containing walls pass neither matter nor energy, the system is said to be isolated and its internal energy cannot change. Microscopically, the internal energy can be analyzed in terms of the kinetic energy of microscopic motion of the system’s particles from translations, rotations, and vibrations, and of the potential energy associated with microscopic forces, including chemical bonds.
Internal energy excludes the kinetic energy of motion of the system as a whole and the potential energy of the system as a whole due to external force fields.
ENERGY, THERMAL
Thermal energy refers to several distinct physical concepts, such as the internal energy of a system; heat or sensible heat, which are defined as types of energy transfer (as is work); or for the characteristic energy of a degree of freedom in a thermal system (kT).
ENTROPY
The word ‘entropy’ comes from the Greek words, `en-tropie’ (intrinsic direction). The concept of entropy came about from investigations into lost energy in heat engines. Entropy was introduced as the concept of ‘transformation-energy’, i.e. energy lost to dissipation and friction. Entropy is a macro state variable for a system that is defined only when the system is in equilibrium.
1789 Count Rumford – heat could be created by friction as when cannon bores are machined.
1803 Lazare Carnot – Losses of moments of activity
1824 Sadi Carnot – Work or motive power can be produced when heat falls through temperature difference
1843 James Joules – expresses the concept of energy and its conservation in all processes. He is unable to quantify the effects of friction and dissipation.
1850s Rudolf Clausius – objected to the supposition that no change occurs in the working body. There is inherent loss of usable heat when work is done, e.g. heat produced by friction. This was in contrast to earlier views, based on the theories of Isaac Newton, that heat was an indestructible particle that had mass.
1877 – Boltzmann – visualized a probabilistic way to measure the entropy of an ensemble of ideal gas particles
Entropy is a state variable. Its thermodynamic definition is “Q/T,” and its statistical mechanics definition is “k ln Ω.” Essential problem in statistical thermodynamics has been to determine the distribution of a given amount of energy E over N identical systems.
FREE ENERGY
The change in the free energy is the maximum amount of work that a thermodynamic system can perform in a process at constant temperature, and its sign indicates whether the process is thermodynamically favorable or forbidden. Free energy is not absolute but depends on the choice of a zero point. Therefore, only relative free energy values, or changes in free energy, are physically meaningful.
(Gibbs function) the thermodynamic function of a system that is equal to its enthalpy minus the product of its absolute temperature and entropy: a decrease in the function is equal to the maximum amount of work available exclusive of that due to pressure times volume change during a reversible, isothermal, isobaric process.
(Helmholtz function) the thermodynamic function of a system that is equal to its internal energy minus the product of its absolute temperature and entropy: a decrease in the function is equal to the maximum amount of work available during a reversible isothermal process.
GAS CONSTANT
The gas constant R is a physical constant that is featured in many fundamental equations in the physical sciences, such as the ideal gas law. It relates the energy scale to the temperature scale, when a mole of particles at the stated temperature is being considered. It is defined as the Avogadro constant multiplied by the Boltzmann constant.
HEAT
A non-mechanical energy transfer with reference to a temperature difference between a system and its surroundings or between two parts of the same system.
In thermodynamics, heat is not the property of an isolated system. It is energy in transfer to or from a thermodynamic system. Heat excludes any thermodynamic work that was done and any energy contained in matter transferred. Heat transfer occurs by the following mechanisms:
(1) Conduction, through direct contact of immobile bodies, or through a wall or barrier that is impermeable to matter.
(2) Radiation between separated bodies.
(3) Convective circulation that carries energy from a boundary of one to a boundary of the other.
(4) Friction due to work done by the surroundings on the system of interest, such as Joule heating.
Quantity of energy transferred as heat can be measured by its effect on the states of interacting bodies. For example, by the amount of ice melted, or by change in temperature of a body in the surroundings of the system.
HEAT, SENSIBLE
Sensible heat is heat exchanged by a body (or thermodynamic system) in which the exchange of heat changes the temperature of the body, and some macroscopic variables, but leaves unchanged certain other macroscopic variables, such as volume or pressure. The term is used in contrast to a latent heat, which is the amount of heat exchanged that is hidden, meaning it occurs without change of temperature.
IDEAL GAS
The volume of ideal gas is essentially made up of electromagnetic substance. At very high pressures and very low temperatures the Ideal gas approximations are not valid.
IDEAL GAS
An ideal gas is a theoretical gas composed of many randomly moving point particles that are not subject to inter-particle interactions. The ideal gas concept is useful because it obeys the ideal gas law, a simplified equation of state, and is amenable to analysis under statistical mechanics.
IDEAL GAS LAW
The ideal gas law is the equation of state of a hypothetical ideal gas. It is often written in an empirical form:
PV = nRT
where P, V and T are the pressure, volume and temperature; n is the amount of substance; and R is the ideal gas constant. It is a good approximation of the behavior of many gases under many conditions, although it has several limitations.
ISOTHERMAL PROCESS
An isothermal process is a change of a system, in which the temperature remains constant: ΔT = 0. This typically occurs when a system is in contact with an outside thermal reservoir (heat bath), and the change in the system will occur slowly enough to allow the system to continue to adjust to the temperature of the reservoir through heat exchange. ΔT = 0 but Q ≠ 0.
LAW (ZEROTH) OF THERMODYNAMICS: TEMPERATURE
If two systems are in thermal equilibrium with a third system, they are in thermal equilibrium with each other.
- This law helps define the notion of temperature.
- Systems in thermal equilibrium with each other have the same temperature.
- Temperature is one-dimensional, that one can conceptually arrange bodies in real number sequence from colder to hotter.
- This law allows the definition of temperature in a non-circular way without reference to entropy, its conjugate variable.
LAW (FIRST) OF THERMODYNAMICS: INTERNAL ENERGY
When energy passes, as work, as heat, or with matter, into or out from a system, its internal energy changes in accord with the law of conservation of energy.
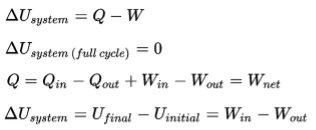
The establishment of the concept of internal energy distinguishes the first law of thermodynamics from the more general law of conservation of energy.
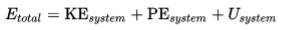
The first law of thermodynamics may be regarded as establishing the existence of the internal energy.
- Equivalently, perpetual motion machines of the first kind are impossible.
A perpetual motion machine of the first kind produces work without the input of energy. It thus violates the first law of thermodynamics: the law of conservation of energy.
- The internal energy of a system is energy contained within the system… It keeps account of the gains and losses of energy of the system that are due to changes in its internal state.
- The internal energy of a system can be changed by transfers: (a) as heat, (b) as work, or (c) with matter.
- When matter transfer is prevented by impermeable containing walls, the system is said to be closed. Then the first law of thermodynamics states that the increase in internal energy is equal to the total heat added plus the work done on the system by its surroundings.
- If the containing walls pass neither matter nor energy, the system is said to be isolated. Then its internal energy cannot change. The first law of thermodynamics may be regarded as establishing the existence of the internal energy.
LAW (FIRST) OF THERMODYNAMICS: INTERNAL ENERGY
When energy passes, as work, as heat, or with matter, into or out from a system, its internal energy changes in accord with the law of conservation of energy.
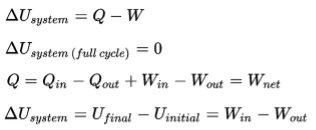
The establishment of the concept of internal energy distinguishes the first law of thermodynamics from the more general law of conservation of energy.
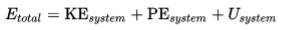
The first law of thermodynamics may be regarded as establishing the existence of the internal energy.
- Equivalently, perpetual motion machines of the first kind are impossible.
A perpetual motion machine of the first kind produces work without the input of energy. It thus violates the first law of thermodynamics: the law of conservation of energy.
- The internal energy of a system is energy contained within the system… It keeps account of the gains and losses of energy of the system that are due to changes in its internal state.
- The internal energy of a system can be changed by transfers: (a) as heat, (b) as work, or (c) with matter.
- When matter transfer is prevented by impermeable containing walls, the system is said to be closed. Then the first law of thermodynamics states that the increase in internal energy is equal to the total heat added plus the work done on the system by its surroundings.
- If the containing walls pass neither matter nor energy, the system is said to be isolated. Then its internal energy cannot change. The first law of thermodynamics may be regarded as establishing the existence of the internal energy.
LAW (FIRST) OF THERMODYNAMICS: INTERNAL ENERGY
When energy passes, as work, as heat, or with matter, into or out from a system, its internal energy changes in accord with the law of conservation of energy.
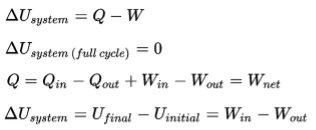
The establishment of the concept of internal energy distinguishes the first law of thermodynamics from the more general law of conservation of energy.
The first law of thermodynamics may be regarded as establishing the existence of the internal energy.
- Equivalently, perpetual motion machines of the first kind are impossible.
A perpetual motion machine of the first kind produces work without the input of energy. It thus violates the first law of thermodynamics: the law of conservation of energy.
- The internal energy of a system is energy contained within the system… It keeps account of the gains and losses of energy of the system that are due to changes in its internal state.
- The internal energy of a system can be changed by transfers: (a) as heat, (b) as work, or (c) with matter.
- When matter transfer is prevented by impermeable containing walls, the system is said to be closed. Then the first law of thermodynamics states that the increase in internal energy is equal to the total heat added plus the work done on the system by its surroundings.
- If the containing walls pass neither matter nor energy, the system is said to be isolated. Then its internal energy cannot change. The first law of thermodynamics may be regarded as establishing the existence of the internal energy.
LAW (SECOND) OF THERMODYNAMICS: ENTROPY
Theorem of the equivalence of transformations
In a natural thermodynamic process, the sum of the entropies of the interacting thermodynamic systems increases.
- Equivalently, perpetual motion machines of the second kind are impossible.
- Indicates the irreversibility of natural processes. NOTE: The precipitation of order from chaos seems to be irreversible.
- When two initially isolated systems in separate but nearby regions of space, each in thermodynamic equilibrium with itself but not necessarily with each other, are then allowed to interact, they will eventually reach a mutual thermodynamic equilibrium. The sum of the entropies of the initially isolated systems is less than or equal to the total entropy of the final combination. Equality occurs just when the two original systems have all their respective intensive variables (temperature, pressure) equal; then the final system also has the same values.
- This statement of the second law is founded on the assumption, that in classical thermodynamics, the entropy of a system is defined only when it has reached internal thermodynamic equilibrium (thermodynamic equilibrium with itself).
- The second law is applicable to a wide variety of processes, reversible and irreversible. All natural processes are irreversible. Reversible processes are a useful and convenient theoretical fiction, but do not occur in nature.
- A prime example of irreversibility is in the transfer of heat by conduction or radiation. It was known long before the discovery of the notion of entropy that when two bodies initially of different temperatures come into thermal connection, then heat always flows from the hotter body to the colder one.
- The second law tells also about kinds of irreversibility other than heat transfer, for example those of friction and viscosity, and those of chemical reactions. The notion of entropy is needed to provide that wider scope of the law.
- Heat Q is proportional to the total kinetic energy (K.E.) of microscopic particles in a system. Temperature T is proportional to the average K.E. of the system. Therefore, the ratio Q/T (entropy) shall be constant for a closed system, being the ratio of total to average K.E. Thus, it would represent the total number of particles in a system. Q reduces as does T when heat energy converts to the mechanical work done.
- Conversion of Heat to mechanical Work is essentially the kinetic energy of microscopic particles converting to kinetic energy of large objects.
- Not all K.E. of microscopic particles can be converted to K.E. of large objects. As long as temperature is not zero (K), the microscopic particles retain some of their K.E.
- When heat is added to a system in which the number of microscopic particles do not change, then both Q and T increase in the same proportion. It is incorrect to assume that T remains constant.
- When Q is converted to mechanical work in a system in which the number of microscopic particles do not change, then both Q and T decrease in the same proportion. It is incorrect to assume that Q remains constant.
Q1 = Q2 + W with a decrease in temperature from T1 to T2.
or, W = Q1 – Q2
or, W = n (T1 – T2), where n is proportional to the number of particles in the system
- Mechanical work done should be proportional to the difference in temperatures in a reversible process. In an irreversible process where losses occur, work w is less than W.
Efficiency = w/W x 100% = w/[n(T1-T2)] x 100%
- Entropy increases when Q remains constant while T decreases. How is Q defined here?
LAW (THIRD) OF THERMODYNAMICS
The entropy of a system approaches a constant value as the temperature approaches absolute zero. The entropy of a perfect crystal of any pure substance approaches zero as the temperature approaches absolute zero.
- At zero temperature the system must be in a state with the minimum thermal energy. This statement holds true if the perfect crystal has only one state with minimum energy.
- The constant value (not necessarily zero) is called the residual entropy of the system.
LAWS OF THERMODYNAMICS
- FIRST LAW: Energy cannot be created or destroyed, only transformed.
MOLE (AVOGADRO CONSTANT)
The mole is the unit of measurement for amount of substance. A mole of particles is defined as 6.022 × 10^23 particles, which may be atoms, molecules, ions, or electrons. The mass of one mole of a chemical compound, in grams, is numerically equal (for all practical purposes) to the average mass of one molecule of the compound, in atomic mass units).
SPECIFIC HEAT CAPACITY
The specific heat capacity of a substance is the amount of energy that must be added, in the form of heat, to one unit of mass of the substance in order to cause an increase of one unit in its temperature. The SI unit of specific heat is joule per kelvin per kilogram, J/(K kg). Liquid water has one of the highest specific heats among common substances, about 4182 J/(K kg) at 20 °C. The specific heat often varies with temperature, and is different for each state of matter.
TEMPERATURE
Temperature is a physical property of matter that quantitatively expresses hot and cold. It is the manifestation of thermal energy, present in all matter, which is the source of the occurrence of heat, a flow of energy, when a body is in contact with another that is colder.
TEMPERATURE
A measure of the warmth or coldness of an object or substance with reference to some standard value.
TEMPERATURE, THERMODYNAMIC
Thermodynamic temperature is defined by the third law of thermodynamics in which the theoretically lowest temperature is the null or zero point. At this point, absolute zero, the particle constituents of matter have minimal motion and can become no colder. Thermodynamic temperature is often also called absolute temperature, for two reasons: the first, proposed by Kelvin, that it does not depend on the properties of a particular material; the second, that it refers to an absolute zero according to the properties of the ideal gas.
THERMODYNAMICS (“force or power of heat”)
The science concerned with the relations between heat and mechanical energy or work, and the conversion of one into the other.
THERMODYNAMIC CYCLE
Every single thermodynamic system exists in a particular state. When a system is taken through a series of different states and finally returned to its initial state, a thermodynamic cycle is said to have occurred. In the process of going through this cycle, the system may perform work on its surroundings, for example by moving a piston, thereby acting as a heat engine.
THERMODYNAMIC EQUILIBRIUM
Macrostates are not changing, but microstates are constantly changing. When a system changes state, the beginning and final states may be well-defined, but the intermediate states may not be defined because the system is not in equilibrium. However, if we can maintain equilibrium the whole time by making the changes very slowly and in very small steps (quasi-static process). Then we can describe the path from beginning and final states. Most quasi-static processes are reversible because there is no loss of energy. In real world there is no perfectly reversible process.
THERMODYNAMIC STATE
A thermodynamic system is a macroscopic object, the microscopic details of which are not explicitly considered in its thermodynamic description.
.
SECTION 4: STATISTICAL MECHANICS
.
BOLTZMANN CONSTANT
It is the proportionality factor that relates the average relative kinetic energy of particles in a gas with the thermodynamic temperature of the gas.
The Boltzmann constant (kB or k), which is named after Ludwig Boltzmann, is a physical constant relating the average kinetic energy of particles in a gas with the temperature of the gas. It is the gas constant R divided by the Avogadro constant NA.
MACROSTATES
Pressure, temperature and volume are macrostates of a system.
MAXWELL-BOLTZMANN DISTRIBUTION
A Maxwell–Boltzmann Distribution is a probability distribution used for describing the speeds of various particles within a stationary container at a specific temperature. The distribution is often represented with a graph, with the y-axis defined as the number of molecules and the x-axis defined as the speed.
MICROSTATES
State of every atom and molecule.
STATE VARIABLE
Internal energy, enthalpy, and entropy are state quantities or state functions because they describe quantitatively an equilibrium state of a thermodynamic system, irrespective of how the system arrived in that state. In contrast, mechanical work and heat are process quantities or path functions, because their values depend on the specific transition (or path) between two equilibrium states.
STATISTICAL MECHANICS
Statistical mechanics involves dynamics, Where the attention is focused on statistical equilibrium (steady state). Statistical equilibrium does not mean that the particles have stopped moving (mechanical equilibrium), rather, only that the ensemble is not evolving.
(1860) Statistical mechanics was developed to define temperature equilibrium… Statistical mechanics shows how the concepts from macroscopic observations (such as temperature and pressure) are related to the description of microscopic state that fluctuates around an average state… Randomness is used to find meaningful information… Discreteness at microscopic scale averages as fluidity on macroscopic scale.
STATISTICAL THERMODYNAMICS (equilibrium statistical mechanics)
The primary goal of statistical thermodynamics is to derive the classical thermodynamics of materials in terms of the properties of their constituent particles and the interactions between them. In other words, statistical thermodynamics provides a connection between the macroscopic properties of materials in thermodynamic equilibrium, and the microscopic behaviors and motions occurring inside the material.
STATISTICS
We can’t keep track of trillions of molecules individually, so we track the average.
.
