According to Wikipedia:
Wave–particle duality is the concept in quantum mechanics that every particle or quantum entity may be described as either a particle or a wave. It expresses the inability of the classical concepts “particle” or “wave” to fully describe the behavior of quantum-scale objects.
A quantum entity is an “energy particle”. This means it is simply the energy involved in an interaction at the atomic level. Examples are photon and electron. Quantum physics then assumes that photon and electron are indivisible particles (see Feynman on Quantum Behavior).
Light cannot be a wave because it is not a disturbance in stationary medium, such as, aether. Nor can it be a particle because it cannot be distinguished in space by a center of mass. As described earlier,
Light is a continuum in space that has a certain density represented by its “frequency”.
.
Double-slit Experiment
According to Wikipedia:
In modern physics, the double-slit experiment is a demonstration that light and matter can display characteristics of both classically defined waves and particles…
In a double-slit experiment with water waves an interference pattern is observed.
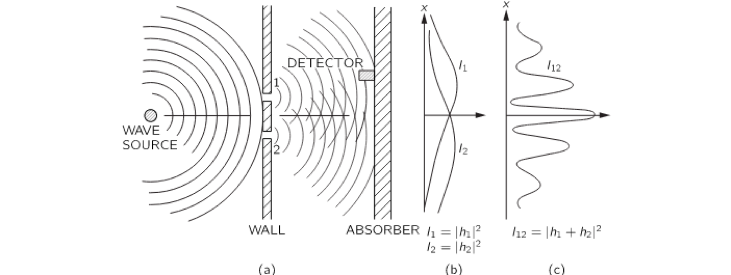
A similar pattern is observed when this experiment is repeated with light instead of water waves. The feature, which is common to both of these instances, is the infinite divisibility of the “flow” arriving at the two slits. In case of the water wave, the disturbance on the surface of water is infinitely divisible being continuous. In the case of light, light itself is an infinitely divisible continuum.
The infinite divisibility of the flow arriving at the double-slit produces the interference phenomenon.
Newton’s corpuscular theory of light considered corpuscles to be infinitely divisible. Therefore, it should never have been dropped in favor of a wave-theory of light that postulated unobserved aether.
.
Assumption in Physics
Physics assumes that light and electron flow arriving at the double-slits are made up of indivisible particles that go through one slit or the other (see Feynman on Quantum Behavior). Quantum mechanics justifies it through the idea of probability but there are no indivisible particles in the first place. Photons and electrons are “energy particles” arising from interactions. They are not indivisible “lumps” in space. The idea of particles is generated as light or electron interacts with the detector in definite amounts to generate clicks.
The wave-particle confusion exists in physics because “energy particles” are thought of as indivisible particles in space.
This is explained in Particle, Continuum and Atom.
.
Quantum Mechanics
Quantum mechanics came about because of the discreteness of energy interactions at the subatomic level.
A material particle also implies discreteness of energy interactions, but, in addition, it implies discreteness of mass in space because of center of mass property. This similarity of energy and mass discreteness does not carry forward from material to subatomic area.
A quantum particle does not imply indivisibility in space.
.

Comments
A substance may be represented by particles or by a fluid-like continuum. When a fluid-like continuum of low density moves rapidly it acquires wave-like characteristics (de Broglie’s hypothesis). This is the case with all non-atomic substances, such as, the electronic flow and the light.
Quantum particles like gluons, bosons or leptons are actually fluid-like continua of different densities that have, primarily, a mathematical significance only. Their discrete mathematical properties come from energy interactions and not from assumed “discreteness in space”. The “particle” in wave-particle property is due the confusion of discrete energy interaction with assumed “discreteness in space”.
Please see above.