Reference: A Logical Approach to Theoretical Physics
The spectrum of quantization describes a gradual coagulation of vis insita, Newton’s innate force of substance, which, ultimately, congeals into the solidity of matter. An atom consists of this entire spectrum. It is a compact structure that forms the smallest unit of matter.
The modern atom is visualized as a highly quantized nucleus surrounded by a barely quantized electronic region. The nucleus is extremely small in size compared to the region surrounding it. The vibrations are so dense in the nucleus that they have the appearance of solidity. The wavelength of vibrations has become so small that the solidity itself forms a continuum. This condition is identified as “mass”.
The atomic unit of mass is the mass of the nucleus of the hydrogen atom. The mass of the nuclei of other atoms are very close to being integer multiples of this unit. This has led to the assumption that the nuclei of atoms are made of particles called “nucleons” that are bound by nuclear force. This assumption is reinforced by nuclear reactions in which nucleons are absorbed or emitted.
This, however, does not exclude the alternate view that all nuclei are single entities in which nucleons get absorbed and emitted. When we say that the nucleus consists of nucleons, it simply means that there are localized regions in the nucleus of still higher frequency that are indistinguishable from one another. There is continuity of substance from one nucleon to another.
Individual nucleons may not exist as separate particles inside the nucleus.
Compared to the substantiality of the mass in nucleus, the substantiality of the surrounding electronic region is almost insignificant. A similar situation exists in the macroscopic view of matter and space, where we overlook the significance of space. This is the basis of “particles in void” framework. This framework is applicable only to the material domain where the substance is made up of atoms; it cannot be applied to radiation. This is the perspective of Newtonian mechanics.
The substantiality of the electronic region in atom is insignificant compared to the mass in the nucleus, but it cannot be ignored in quantum mechanical studies of the interface between the nucleus and electronic region. It is conjectured here that the charge between these two regions exist only because of the extremely high gradient of substantiality between them. An electron expelled from the atom is still bound to a positive nucleus. Hopefully, I shall be able to prove this conjecture soon.
In current physics, the electronic region is assumed to be made up of particles called “electrons” because mathematical calculations yield integral relationships, and also because this region absorbs and emit electrons. The electronic region being continuous cannot consist of discrete electron particles. This continuity is acknowledged by Schrödinger in his study of the electronic region.
Electrons are “particles” in the sense that they are radiation of high substantiality in the gamma region of the spectrum.
The Schrödinger’s equation1 was successfully solved for the emission of light from a hydrogen atom. The solution agreed with Bohr’s quantized energy levels. It even provided the energy levels observed, which could not be predicted from the Bohr’s model. It was a considerable advance to have determined these energies by a wave-theory instead of by an inexplicable mathematical rule. The Schrödinger’s model defines the point location of the subatomic particle by a probability distribution function. The concept of “probability” may be given a more realistic meaning using the “continuum of substance” framework. Hopefully, this will improve the workability of this equation for more complex atoms.
The success of Schrödinger’s wave-theory, however, puts the particle model of atom in doubt.
It is very likely that an atom is a homogeneous entity with no discrete particles existing inside it. The interactions with the atom may suffice to generate electrons, protons and neutrons. If we do not assume subatomic particles to reside within an atom, we can express the atomic structure in terms of rapidly quantizing whirlpool of radiation.
The atom may be modeled as a “sink” for quantizing radiation. This means that the atom provides a location where radiation may condense and terminate as mass. There are threshold frequencies at which radiation converges into the atom and then forms the nucleus at the center of the atom.
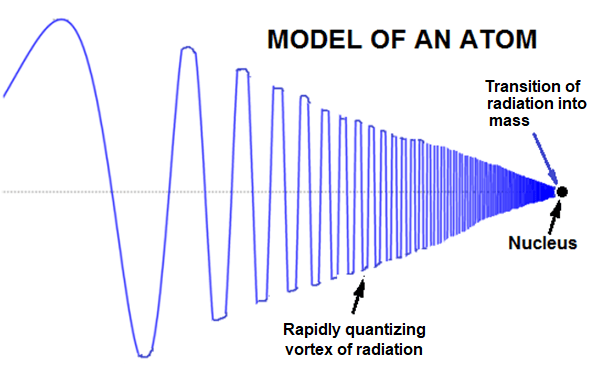
This model may be identified as the “whirlpool” model of the atom.
The Bohr’s model of atom has helped provide insight into the Periodic Table; but, it soon becomes very complex when describing the atomic structure beyond the simplest hydrogen atom. Hopefully, the “whirlpool” model of atom shall provide deeper insight into the structure of the atom with simpler math.
In a blackbody, the atomic configurations consist of “oscillators” over the whole range of frequency spectrum. When a blackbody is heated, it emits radiation at all frequencies. Oscillators of higher frequencies require increasing energy to be activated.
We may assume “oscillators” in the electronic region of the atom that, when irradiated, become activated and emit characteristic radiation.
Atoms are stable configurations of extremely high quantization levels in space. A free electron is a ray of highly substantial radiation.
Schrödinger’s wave-theory may be improved doing away with the particle model of the atom.
.
1”The Nature of The Physical World” by A. S. Eddington, Chapter X (section 4)
.
