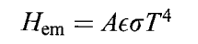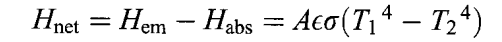Reference: Beginning Physics I
CHAPTER 17: TRANSFER OF HEAT
.
KEY WORD LIST
Conduction, Conductors, Insulators, R-Factor, Convection, Radiation, Stefan-Boltzmann Law, Emissivity, Blackbody
.
GLOSSARY
For details on the following concepts, please consult CHAPTER 17.
CONDUCTION
Heat conduction is the direct transfer of thermal energy from one layer of molecules to the next layer. The amount of transferred per unit time across a given cross section of a bar is directly proportional to the temperature difference and to the area and is inversely proportional to the length.
The proportionality constant in this equation is different for each material. It is called the coefficient of thermal conductivity (or conductivity, for short).
CONDUCTORS
Metals generally have larger conductivities than other solids and are therefore called good heat “conductors.”
INSULATORS
Materials that clearly don’t conduct heat well are called insulators.
R-FACTOR
The R-factor of a slab is its length divided by conductivity: R = L/k
Therefore, we have, H/A = ∆T/R, where R = R1 + R2 + R3
CONVECTION
Convection is a mechanism for the transfer of thermal energy that applies to fluids (liquids and gases). Unlike conduction, where there is no macroscopic migration of molecules, in convection the thermal energy is transferred by the motion of material from one place to another.
To a good approximation the rate of convective heat flow is proportional to the area of the contact surface and to the temperature difference between the surface and the bulk of the fluid away from the surface.
H = h A ∆T
where h, the coefficient of convection, depends on the fluid, the geometry, and a variety of other factors (including a slight dependence on ∆T).
If the circulation of the fluid is aided by a fan or pump, it is called forced convection. If the circulation is the consequence of the natural difference in density of the fluid (caused by a temperature difference) at different locations, it is called natural convection.
RADIATION
Radiation is a process that involves electromagnetic waves. Every substance at any temperature emits electromagnetic radiation, which carries energy with it. For a system to be in thermal equilibrium with its surroundings it must absorb as much radiation as it emits.
STEFAN-BOLTZMANN LAW
The total amount of radiation energy emitted per second from an object at uniform temperature and surface area is,
Where ϵ is a dimensionless constant, called the emissivity, with a value between 0 and 1 that varies from substance to substance; and σ is a universal constant called the Stefan-Boltzmann constant with the value
For an object at temperature T1 enclosed in a container with walls at temperature T2, the net rate of flow of thermal energy out of the object is
EMISSIVITY
The emissivity is a dimensionless constant with a value between 0 and 1 that varies from substance to substance. The emissivity is 1 for a good emitter. A good emitter is also a good absorber. At normal temperatures a good absorber-emitter appears black.
BLACKBODY
A perfect or ideal absorber-emitter (e = 1) is called a blackbody, but no real object is a perfect blackbody.
.