Reference: Disturbance Theory
.
-
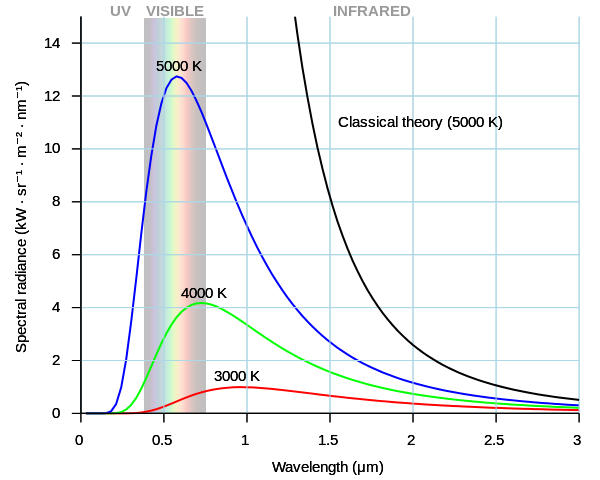
The Classical Mechanics made a transition into Quantum Mechanics at the beginning of 20th century when the interactions between field and matter were studied. The first field-matter interaction was encountered in the Black Body Radiation. The classical equipartition theory failed to account for the energy of the emitted electromagnetic spectrum.
-
There was a thermodynamic equilibrium observed between the temperature of the body and the spectrum of the electromagnetic field surrounding the body. In other words, the agitation of atoms (temperature) was in equilibrium with the absorption and emission of thermal electromagnetic radiation (spectrum).
-
The formulae based on classical thermodynamics could either explain the low frequency part of the spectrum (Raleigh-Jean formula), or the high frequency part of the spectrum (Wien’s Distribution formula), but not the entire spectrum at once. Planck found the formula, which could replicate the entire spectrum by ingeniously interpolating between the above two formulae. This was purely an empirical effort based on mathematics. He came up with the explanation for his formula later.
-
From Derivation of Planck’s radiation law:
In order to reproduce the formula which he had empirically derived and presented in October 1900, Planck found that he could only do so if he assumed that the radiation was produced by oscillating electrons, which he modelled as oscillating on a massless spring (so-called “harmonic oscillators”). The total energy at any given frequency would be given by the energy of a single oscillator at that frequency multiplied by the number of oscillators oscillating at that frequency.
However, he had to assume that
- The energy of each oscillator was not related to either the square of the amplitude of oscillation or the square of the frequency of oscillation (as it would be in classical physics), but rather just to the frequency,
E α ν - The energy of each oscillator could only be a multiple of some fundamental “chunk” of radiation, hν, so En = nhν
where n = 0, 1, 2, 3, 4 - The number of oscillators with each energy En was given by the Boltzmann distribution, so
Nn = N0e–nhν/kT
where N0 is the number of oscillators in the lowest energy state.
By combining these assumptions, Planck was able in November 1900 to reproduce the exact equation which he had derived empirically in October 1900. In doing so he provided, for the first time, a physical explanation for the observed blackbody curve.
- The energy of each oscillator was not related to either the square of the amplitude of oscillation or the square of the frequency of oscillation (as it would be in classical physics), but rather just to the frequency,
-
The frequency of the radiation matched the frequency of the “oscillators” in the body. The high frequency oscillators could be activated only when energy proportional to their frequency was available. Therefore, lesser numbers of oscillators were activated at higher frequencies. Planck thus resolved the Ultraviolet catastrophe.
-
We may postulate that the kinetic and potential states of oscillators produce the electric and magnetic states of radiation respectively. Therefore, the electric state may be related to magnetic state the way the kinetic state is related to potential state. The magnetic state could be a concentrated electric state; and the electric state could be a flowing magnetic state.
-
Thus an electromagnetic cycle consists of a pulse of energy of magnitude ‘h’. A three-dimensional electromagnetic field is made up of such dynamic pulses.
.