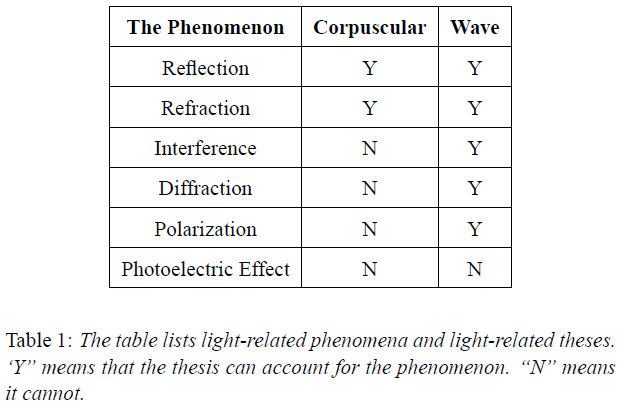
Reference: Spacetime 5: A New Model of Atom
The section Outline of Schrodinger’s Theory from Eddington’s book, “The Nature of the Physical World,” provides a wonderful description of Schrodinger’s model that may be compared to the Disturbance theory.
According to Schrödinger there is a sub-aether, which is rippling at a very high frequency. These ripples converge and coalesce to generate disturbed areas in space that act as discrete particles, such as, electrons.
This is very much in line with the Disturbance theory, except that Schrödinger’s sub-aether is identified as space. The ripples in space result in a frequency spectrum that spans from undisturbed space of zero frequency at the lower end to extremely high frequencies at the upper end that collapse into mass. The very high frequency ripples of Schrödinger’s model are identified as the gamma ray region of the electromagnetic spectrum.
In Schrödinger’s model the velocity of ripples varies with wavelength or period. Those of shorter period travel faster. The speed may also be modified by local conditions, which may be compared to field of force.
In Disturbance theory, the speed of propagation is meaningless in subatomic regions where the constitution of space and time itself is changing because of relativistic effects, though space/time maintains a constant ratio ‘c’. The monitoring variable, therefore, is not speed but frequency.
The stormy regions of Schrödinger’s model are high frequency regions that are viewed as particles. In the Disturbance model they are viewed as 3-D “whirlpools” existing in a low frequency background. The high frequency gradients defining them influence the surrounding low frequency region.
The Schrödinger’s Equation is based on the idea of conservation of energy. The terms of the Schrödinger equation can be interpreted as total energy of the system, equal to the system kinetic energy plus the system potential energy. This equation is solved for the motion of sub-atomic particles. Frequency is recognized as the energy of the particle, and it provides the relationship between period and energy per the h rule. The motion of the Schrödinger’s particle is represented by the group-velocity and not the wave-velocity.
In Disturbance theory, the 3-D whirlpools are not viewed as particles. Instead they are made up of spherical shells. Frequency is associated with the excitation energy of these shells. Each cycle of this frequency has an energy equal to the Planck’s constant h. The excitation energy for these shells is represented by the frequency of light absorbed or emitted, and not by the high frequency that make up the shells.
The Schrödinger’s model defines the point location of the subatomic particle by a probability distribution function. This is not necessary for the shell represention of the model in Disturbance theory. The shells are made up of high frequency waves. They do not constitute a point particle. The only particle is the nucleus of the atom where extremely high frequency waves collapse as mass.
The Schrödinger’s equation was successfully solved for the emission of light from a hydrogen atom. The nucleus was represented by a “field of force” (potential energy) that influenced the motion of the electron (kinetic energy). The solution was a discrete set of frequencies that described the possible states of the electron. It agreed with Bohr’s quantized energy levels. It even provided the energy levels observed, which could not be predicted from the Bohr’s model. It was a considerable advance to have determined these energies by a wave-theory instead of by an inexplicable mathematical rule.
Eddington says, “It would be difficult to think of the electron as having two energies (i.e. being in two Bohr orbits) simultaneously; but there is nothing to prevent waves of two different frequencies being simultaneously present in the sub-aether. Thus the wave-theory allows us easily to picture a condition which the classical theory could only describe in paradoxical terms.” Light emitted from an atom is the difference between two energy levels of the electron. This is viewed in Schrödinger’s theory as the “beat” produced by two waves that are close to each other in frequency as in heterodyning.
This places the particle model of electron in doubt. This problem is not there in the Disturbance theory where light emission may be explained as “beats” produced by two adjacent oscillating shells.
Schrödinger assumes a wave function ψ in sub-aether as an elementary indefinable of the wave-theory. The probability that the particle or electron is within a given region is interpreted as being proportional to ψ2 in that region.
In Disturbance theory, the atomic structure consists of shells whose frequencies are in the gamma range. These frequencies increase as one moves closer to the center of the atom. These “shells” oscillate when excited. Localization occurs only in terms of the shell that is oscillating. Two closely resonating shells have “beats”that appear as light absorbed or emitted.
An oscillating shell may represent an electron inside the atom. As shells oscillate in succession, the electron may appear to move. The concept of “probability” in Schrödinger’s model may thus be given a meaning through Disturbance theory.
The “shells” increase in frequency as they get closer to the center until they “collapse” to form the nucleus. In a nucleus, these shells are so close together that they approach the classical definition of a particle.
The picture of electron as a classical particle is much less proper compared to the nucleus. This lack of propriety is expressed indirectly by Heisenberg’s uncertainty principle.
Schrödinger’s sub-aether requires six-dimensions to describe two electrons within an atom, thus the sub-aether does not exist in physical space. There is no such problem with the model per Disturbance theory, which does not require additional dimensions to describe two electrons.
Summary
The sub-aether of Schrödinger is an arbitray concept that is not consistent with real space. It becomes complex very rapidly as more electrons are considered. There is no sub-aether. There is simply the physical space. Disturbances in this space are adequately described by the broad electromagnetic spectrum. Schrödinger’s very high frequencies of sub-aether are better described by the gamma range of the electromagnetic spectrum.
In reality, there is no electron, as a particle, possible within the atom that can assume two different energies simultaneously. Instead of particles there are cascading spherical shells in the atom with frequencies in the gamma range. These frequencies increase toward the center of atom. Instead of electrons there are oscillating shells. When oscillating, some adjacent shells produce lower frequency “beats”, which appear as light absorbed or emitted by the atom.
There are no electrons as particles in atom. There are only high frequency shells that respond to excitation.
.