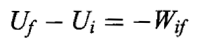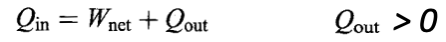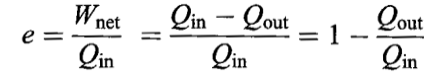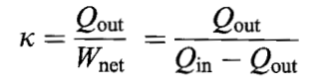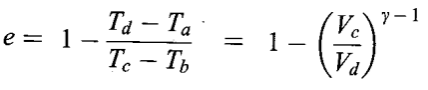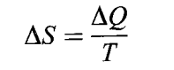Reference: Beginning Physics I
CHAPTER 18: THE FIRST & SECOND LAWS OF THERMODYNAMICS
.
KEY WORD LIST
The First Law of Thermodynamics, P-V Diagram, Quasistatic Processes, Cyclical Process, Isochoric Process, Isobaric Process, Isothermal Process, Adiabatic Process, Carnot Cycle, The Second Law of Thermodynamics, The Engine Statement of The Second Law, Efficiency, The Refrigerator Statement of The Second Law, Co-Efficient of Performance, Carnot Engine, Otto Cycle, Compression Ratio, Entropy, Entropy of The System
.
GLOSSARY
For details on the following concepts, please consult CHAPTER 18.
THE FIRST LAW OF THERMODYNAMICS
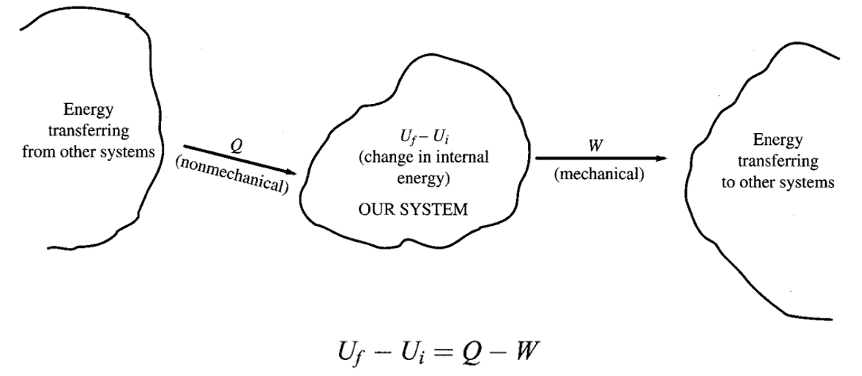
The first law of thermodynamics is the statement of the law of conservation of energy in its most general form. It presumes that the overall energy of the universe remains the same.
If Ui and Uf are the initial and final total internal energy of the system, respectively, at the beginning and at the end of the process, we must have
Where Q is the algebraic heat entering the system; and W is the algebraic work done by the system on the outside world during the same process.
In other words, energy can only shift from one system to another (by means of work and heat transfer), but the total energy of the universe stays fixed.
P-V DIAGRAM
For a quasistatic process, the evolving states of the system can be tracked as a path on a P-V diagram. The work done by the system between the states i and f is the total area under the curve on the P-V diagram.
Reversing a given quasistatic path reverses the sign of the work done and the heat transferred.
QUASISTATIC PROCESSES
The quasistatic processes include a constant-volume (isochoric) process, a constant-pressure (isobaric) process, a constant-temperature (isothermal) process, and a process in which no heat enters or leaves the system (adiabatic process).
CYCLICAL PROCESS
A cyclical process may consist of the same of different quasistatic processes. In a cyclical process the work done is plus or minus the area enclosed by the closed cycle path on the P-V diagram.
ISOCHORIC PROCESS
In an isochoric process the work performed is zero. Therefore, the first law of thermodynamic may be expressed for an isochoric process as,
ISOBARIC PROCESS
The first law of thermodynamic may be expressed for an isochoric process as,
ISOTHERMAL PROCESS
For an ideal gas, in an isothermal process, the internal energy at every point along an isotherm is the same.
ADIABATIC PROCESS
For an adiabatic process the first law takes the form,
For an ideal gas undergoing the adiabatic process, pressure and volume are related by
CARNOT CYCLE
A Carnot cycle is a system undergoing a quasistatic cyclical process involving four legs, with two being isotherms and two being adiabats. Such a process is represented on the P-V diagram as follows:
THE SECOND LAW OF THERMODYNAMICS
The second law of thermodynamics addresses the question of the feasibility of certain types of energy transfers. To accomplish the removal of thermal energy from a cool body and transfer it to a hot body requires an intermediary system called a refrigerator. To convert thermal energy to mechanical energy requires the services of an intermediary system called a heat engine. These intermediary systems effect the transfer that do not occur naturally. The second law is deeply connected to the concept of randomness, and therefore to the subject of statistical mechanics.
THE ENGINE STATEMENT OF THE SECOND LAW
“It Is Impossible for a cyclical process to have no other effect than to draw thermal energy from some system(s), and to convert it completely into mechanical energy.”
EFFICIENCY
The efficiency of any engine is defined as the ratio of mechanical energy obtained to the thermal energy extracted from the hot reservoir.
THE REFRIGERATOR STATEMENT OF THE SECOND LAW
“It is impossible for a cyclical process to have no other effect than to extract thermal energy from a cooler system and eject that thermal energy to a hotter system(s).”
CO-EFFICIENT OF PERFORMANCE
The co-efficient of performance of a refrigerator is defined as.
CARNOT ENGINE
The second law implies that the most efficient engine operating between two fixed temperature reservoirs is a Carnot engine. The efficiency of a Carnot engine is,
OTTO CYCLE
One cylinder of a gasoline engine can be idealized by a quasistatic engine called the Otto cycle, as shown below.
The efficiency of this cycle is,
COMPRESSION RATIO
The compression ratio is the ratio of the largest volume to the smallest volume of the engine cylinder as the piston moves in and out. The greater is the compression ratio, the more efficient is the engine.
ENTROPY
For every equilibrium state of a system there is a definite quantitative measure of the disorder of the system in that state. This quantitative measure assigns a value to each equilibrium state of the system, which is called entropy. The incremental change in the entropy of a system when a small amount of heat is slowly added, is
The second law of thermodynamics can be restated in terms of the overall entropy of the universe: In any process or interactions of systems, the overall entropy change of the universe obeys,
where the equality occurs only in the case of quasistatic processes.
ENTROPY OF THE SYSTEM
The macroscopic equilibrium state of a system corresponds to the most probable system state with its specific value of the number of ways that the microscopic variables can arrange themselves so as to produce the value of the macroscopic variables that characterize the equilibrium state. The entropy of the system is formally defined as,
where k is Boltzmann constant and Γ is the number of ways.
.