Reference: Beginning Physics I
CHAPTER 13: FLUIDS AT REST (HYDROSTATICS)
.
KEY WORD LIST
Hydrostatics, Density, Specific Gravity, Pressure, Hydrostatic Pressure, Gauge Pressure, Hydraulic Press, Open-Tube Manometer, Barometer, Archimedes’ Principle, Surface Tension, Capillarity
.
GLOSSARY
For details on the following concepts, please consult CHAPTER 13.
HYDROSTATICS
Origin: “still water.” Hydrostatic is that branch of physics that deals with the static fluids, usually confined to the equilibrium and pressure of liquids.
DENSITY
The density d of any substance is defined as the mass per unit volume of the substance. If we have a uniform sample of materials (solid, liquid, or gas) of mass M and volume V, then
SPECIFIC GRAVITY
The specific gravity of a substance is defined as the ratio of the density of the substance to that of water.
PRESSURE
The pressure P on any surface is defined as the force per unit area acting perpendicular to that surface
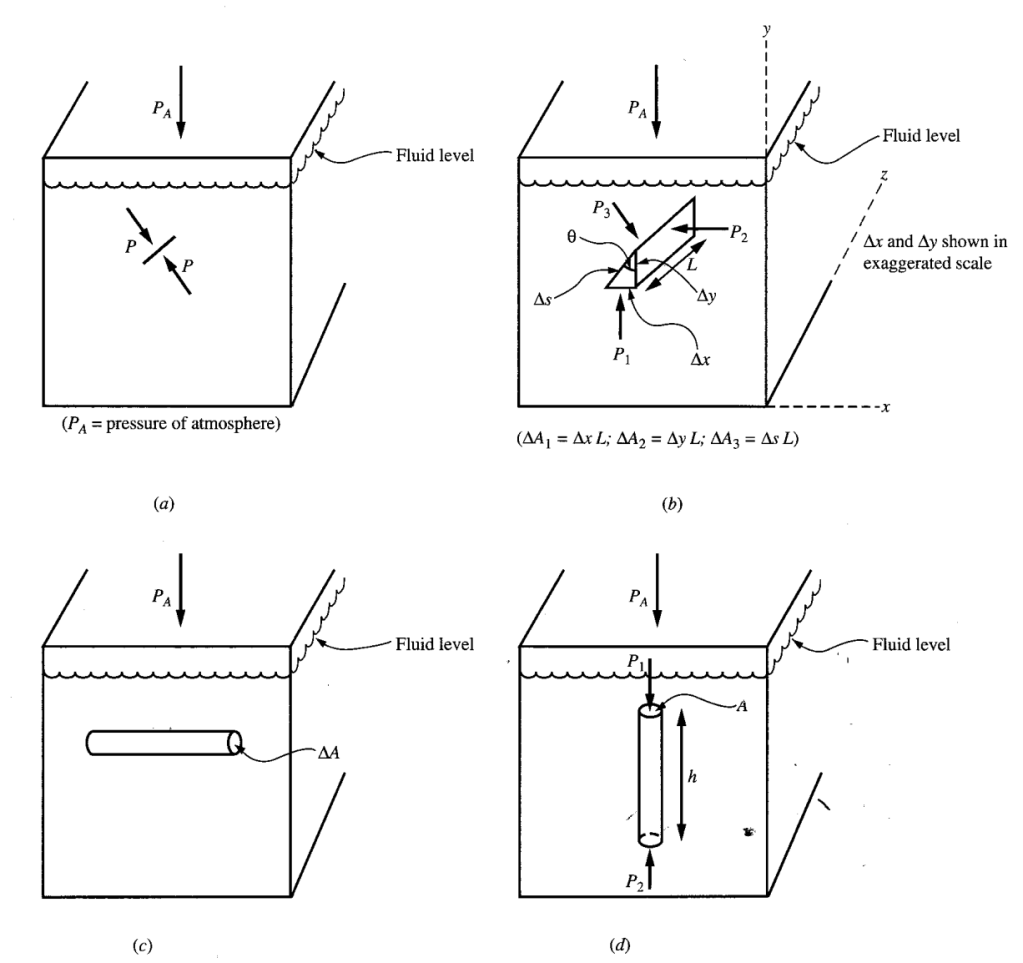
HYDROSTATIC PRESSURE
The hydrostatic pressure is the pressure in a fluid at rest.
- For any point in a fluid at rest, the pressure on one side of a small surface is the same as the pressure on other side.
- The pressure at a given point in the fluid at rest has a definite value that represents the force per unit area on a small surface placed at that point, oriented in any arbitrary direction.
- The pressure in a fluid at rest is the same at all points on a horizontal plane.
- The pressure in a fluid at rest varies only with the depth in accord with the equation,
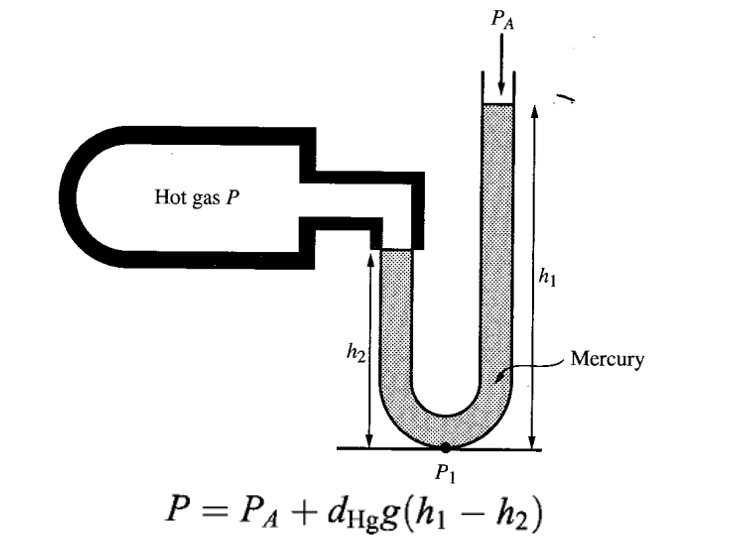
GAUGE PRESSURE
The gauge pressure is the difference between the actual, or absolute pressure P in a fluid, and the pressure exerted by the atmosphere PA, which pervades the surface of the earth.
HYDRAULIC PRESS
OPEN-TUBE MANOMETER
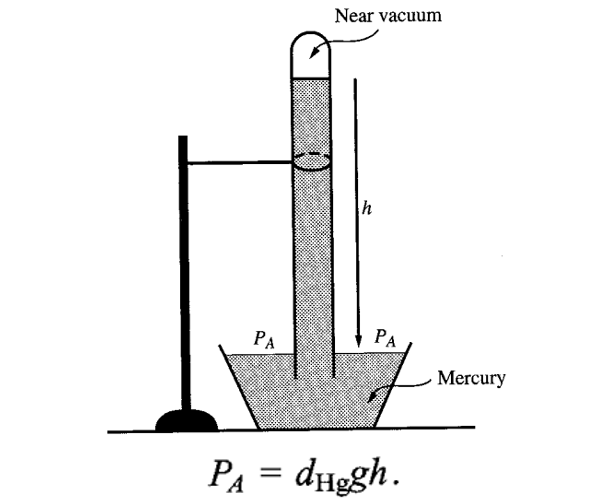
BAROMETER
ARCHIMEDES’ PRINCIPLE
The fact that the buoyant force equals the weight of the displaced liquid is called the Archimedes’ Principle: the law of buoyancy.
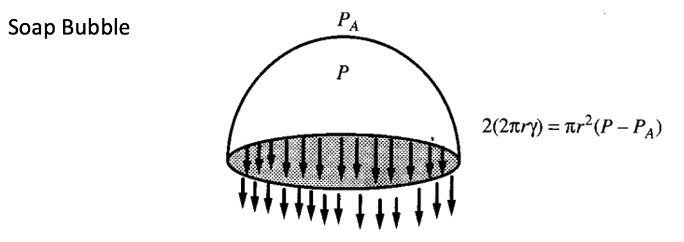
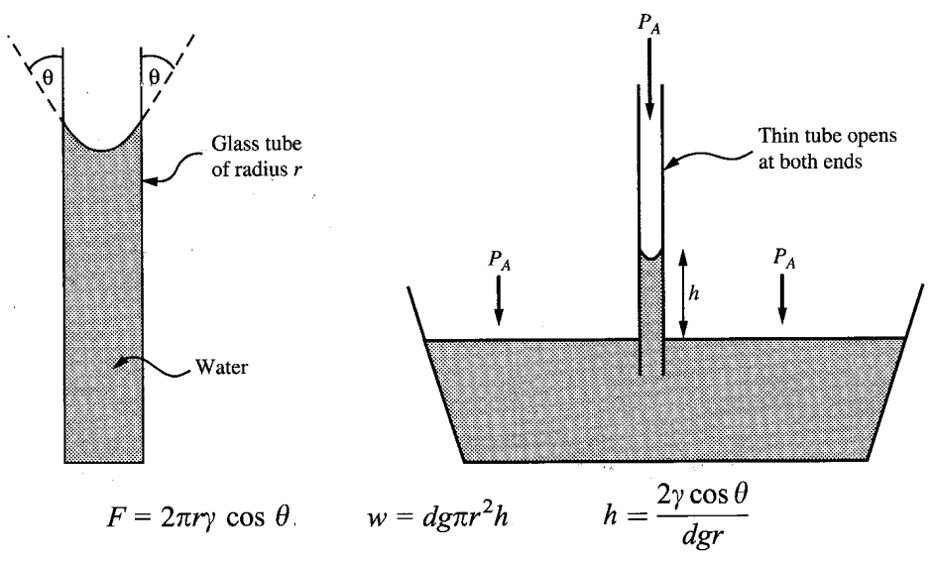
SURFACE TENSION
Surface tension is caused by the pull of the molecules below the surface of the liquid on the molecules at the surface. This tends to pull the surface into a smooth and compact layer. The surface tension is defined as force per unit length exerted by a liquid surface on an object, along its boundary of contact with the object. This force is parallel to the liquid surface and perpendicular to the boundary line of contact. For a straight boundary of length L and a total force F we have
CAPILLARITY
Because of adhesion, the water surface gets pulled toward the wall of the container and bends upward at the point of contact.
.