Reference: Propagation of Light
When space is disturbed electric and magnetic fields are generated. These fields oscillate as a transverse wave. The disturbance travels in a straight line at the speed of light.
Disturbance is manifested in space as oscillating electric and magnetic fields.
In each oscillation the disturbance advances by a distance called wavelength. The oscillations occur at a certain frequency. The speed of propagation is then determined by the product of frequency and wavelength. Oscillations are the result of a rotating vector. The propagation of disturbance may be compared to the advance of a rotating screw.
The disturbance propagates like a rotating vector threading into the space.
The space does not provide any resistance. The disturbance contains its own inertia through the interaction between electric and magnetic fields. This inertia resists any change to frequency and wavelength, and keeps the disturbance going.
The disturbance consists of its own inertia that sustains it.
The disturbance has a large range of frequencies and wavelengths. This range is represented by the electromagnetic spectrum consisting of radio waves, microwaves, infra-red light, visible light, ultra-violet light, X-rays and Gamma rays. This spectrum presents increasing frequency and shortening wavelengths, while the speed of propagation remains practically the same.
The disturbance forms a spectrum of increasing frequency and shortening wavelengths.
Since the spectrum extends over a very large range of frequencies, it may be managed more conveniently as Disturbance Levels on a logarithmic scale of base 2 (similar to octaves).
Disturbance Level Frequency
0 20 or 1
1 21 or 2
2 22 or 4
3 23 or 8
… …
n 2n
The disturbance levels of some of the electromagnetic frequencies are as follows
EM Frequency Disturbance Level
Visible light ………………….. ~ 49
Gamma Rays ………………… ~ 65
Electron ………………………. 66.7
Proton ………………………… 77.6
Neutron ……………………….. 77.6
Inertia seems to increase with increasing disturbance levels. The disturbance levels may provide a measure of inertia.
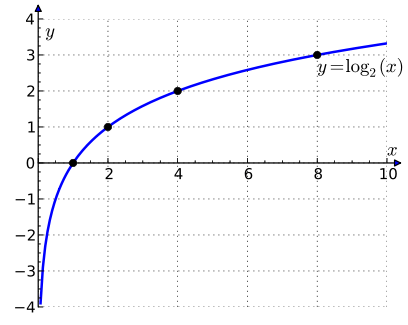
x represents frequency; y represents Disturbance Levels
These disturbance levels may be plotted as above. It can be seen from this graph that negative disturbance levels may be postulated to exist with the halving of frequency. The frequency never reaches zero, except theoretically. We may postulate a level of zero frequency to describe undisturbed space. This space forms the background of all disturbance. The undisturbed space has zero inertia.
The background of all phenomena is undisturbed space of zero frequency and no inertia.
The disturbance levels express inertia. When disturbance level crosses a threshold, the inertia transitions from “wave-disturbance” to “particle-mass.” Inside the atom, this transition occurs at the boundary of the nucleus.
The concept of “Disturbance Level” shall be used in subsequent discussions.
.


Comments
Asimov: “Since electrons do not, in actual fact, spiral into the nucleus, another model must be found. Such a model must account not only for the fact that atoms radiate light (and absorb it. too) but that they only radiate and absorb light of certain characteristic wavelengths (or frequencies). By studying the interrelationships of these characteristic wavelengths, hints may be found as to what that structure might be. Hydrogen would be the element to tackle, for it produces the simplest and most orderly spectrum.”
.
The characteristic wavelength’s may give some indication of the disturbance levels of various shells around the nucleus.
.
Asimov: “Bohr suggested that the electron had a certain minimum orbit, one that represented its ground stale; at which time it was as close to the nucleus as it could be, and possessed minimum energy. Such an electron simply could not radiate energy (though the reason for this was not properly explained for over a decade). Outside the ground state were a series of possible orbits extending farther and farther from the nucleus. Into these orbits, the excited states, the electron could be lifted by the absorption of an appropriate amount of energy.”
.
Instead of orbits, there will be spherical shells in the Disturbance model of the atom. These shells would be such that they would accommodate an integer number of “threads” or wavelengths. They would rather be described by their disturbance levels. Beyond the ground state shell the wave-frequency type inertia shall collapse into particle-mass type inertia. The math for this may be similar but it needs to be worked out.
.
Asimov: “Bohr arranged the orbits about the nucleus of the hydrogen atom in such a way as to give the electron a series of particular values for its angular momentum. This momentum had to involve Planck’s constant since it was that constant that dictated the size of quanta.”
.
Disturbance shells will have orbital momentum around the center of the atom, instead of angular momentum as electrons orbit around in Bohr’s model. Here math will differ.
.
Asimov: “So far, so good. If the lines of the hydrogen spectrum had been simple lines, the Bohr model of the hydrogen atom might have been reasonably satisfactory. However, as spectral analysis was refined, it turned out that each line had a fine structure; that is, it consisted of a number of distinct lines lying close together. It was as though an electron dropping down to orbit 2, for instance, might drop into any of a number of very closely spaced orbits.
This threatened the quantum interpretation of the atom, but in 1916 the German physicist Arnold Sommerfeld (1868-1951) offered an explanation. Bohr had pictured the electron orbits as uniformly circular, but Sommerfeld suggested they might also be elliptical. Elliptical orbits of only certain eccentricities could be fitted into the quantum requirements, and for any principal quantum number, a fixed family of orbits-one circular and the rest elliptical–was permissible, the angular moments among the orbits being slightly different. A drop to each of the various members of the family produced radiation of slightly different frequency.”
.
Ultimately, four quantum numbers (principle, l, m, n) are required to explain the spectral lines from a hydrogen atom. These are defined in terms of ground versus excited states, circular versus elliptical orbits, tilt of the orbits by various amounts, and spin of the electron.
This complication arises because electrons are assumed to be particles orbiting around the nucleus similar to planets orbiting around the sun. This may not be the case.
Instead of planar orbits of electrons, there could be spherical shells of disturbance of increasing disturbance levels as one moves closer to the nucleus.
Math could be worked out for either model that explains the experimental spectral lines. This shows how math could be dependent on the model conceived. Math by itself cannot be trusted. The more complex math is required, the more suspicious one should become about the model. This is one application of Occam’s razor.
.
I have modified the graphics in the OP.
The top graphic may be modified to show the Disturbance model of the atom in which the wave-frequency form of inertia shall dominate the inetrior of the atom; and a small dark spot at the center shall represent the nucleus. The disturbance levels shall be increasing in the direction toward the nucleus. The nucleus shal be particle-mass form of inertia. The surface of the nucleus shall operate as the threshold at which inertia transitions from wave-frequency to particle-mass.
The elctrons shall form out of the outer surface of the atom. This is where chemical interactions shall also take place. All observed phenomenon should be explainable through this model.
Asimov: “While the notion of electron shells and sub-shells finally rationalized the periodic table, even down to the until-then puzzling lanthanides, the Bohr model itself, even as modified by Sommerfeld and others, did not stand up in its original form. The attempt to produce a literal picture of the atom as consisting of electron particles moving in orbits that were circular, elliptical and tilted– much more complicated than the solar system, but still with some key points of similarity– grew top-heavy and collapsed.
During the early 1920’s, it became more common to think, not of orbits, but of energy levels. Electrons moved from one energy level to another and the difference in energy levels determined the size of the quantum (hence the frequency of the radiation) emitted or absorbed.”
.
Energy-level model is closer to the Disturbance-level model, however it seems that old quantum numbers for supposedly embedded electrons are still being used.
.
Asimov: “In the case of a single atom, energy levels could be pictured as simple lines at given heights above the base of a schematic drawing. Two electrons of opposite spin could occupy any of the energy levels and could shift from one level to any other that was not fully occupied. The spaces between the lines represented “forbidden gaps” within which no electron could be located. Each element had its own characteristic collection of lines and gaps, of course.”
.
Energy-levels should be relegated to the atom as a whole and not to “electrons” in the atom.
.
Asimov: “If two atoms of an element are in close proximity, the picture becomes more complicated. The outer electrons of the two atoms are close enough for the energy levels to merge. For each energy level, the electron population is doubled. An energy level cannot hold more than its capacity (two electrons of opposite spin); consequently, what happens is that the energy levels associated with the two atoms shift a bit—one becoming slightly higher than the other. Each can then hold its own electrons.”
.
Trying to describe energy-levels in terms of electrons is making math unnecessarily complex. Hopefully, the math based on Disturbance levels will be simpler.
.
Are the surface energy or disturbance levels of all atoms about the same? Do these disturbance levels only get higher inside the atom? This means that the diturbance levels near the center of the atom shall increase with the atomic mass.
There seems to be a relationship of a disturbance level inside the atom with its distance from the center of the atom (r) and radius of the inertial transition surface (radius of the nucleus, R).
DL = f (r, R)
The radius of the inertial transition surface may depend on the atomic mass (M) of the atom.
R = f (M)
The atomic mass has discrete values, which are currently represented by the sum of protons and neutrons in the nucleus..
DL may also assume discrete values depending on how its wavelength fits in the spherical shell around the center of the atom.
.
It appears that what is currently considered an electron inside the atom is simply a DL with a discrete radius, or an energy level, having a discrete value.
I expect there to be a distinct spectral line for every “electron” inside the atom. This observation shall depend on very fine resolution.
Asimov: “Bohr’s application of quantum theory to atoms had thus proven incalculably fruitful, both in theory and application. Not only was the periodic table rationalized but a whole realm of solid-state devices had grown out of it. Physicists had every reason to be delighted with the results.”
.
It appears that the unexcited configurations of disturbance levels shall be more stable one. These states shall be closer to the nucleus. When the atom gets excited, the disturbance levels move away from the nucleus. Later they settle down closer to the nucleus again after emitting a photon, and losing the excitation energy.
It seems that as disturbance level moves away from the nucleus its wavelength increases, and so does its speed. The excitation energy contributes to this increase in speed. The speed is, therefore, variable but this speed cannot exceed c (speed of light). Hence the speed of disturbance level inside the atom is less than c (speed of light).
The disturbance levels increase as the electromagnetic wave gets more condensed inside the atom. This seems to be accompanied by decrease in speed from c outside the atom. Thus, the speed of waves in the electromagnetic spectrum would depend on their disturbance level. This speed remains pretty constant at c, until the disturbance levels become very high as in case of gamma rays. Thus, at the upper end of the electromagnetic spectrum, the speed shall decrease rapidly from c with increasing frequency of disturbance levels.
This makes sense because the inertia starts to increase rapidly toward the upper end of the spectrum, until it crosses the threshold of transition into particle-mass.
.