Reference: The Nature of the Physical World
This paper presents Chapter X (section 1) from the book THE NATURE OF THE PHYSICAL WORLD by A. S. EDDINGTON. The contents of this book are based on the lectures that Eddington delivered at the University of Edinburgh in January to March 1927.
The paragraphs of original material are accompanied by brief comments in color, based on the present understanding. Feedback on these comments is appreciated.
The heading below links to the original materials.
.
The New Quantum Theory
The conflict between quantum theory and classical theory becomes especially acute in the problem of the propagation of light. Here in effect it becomes a conflict between the corpuscular theory of light and the wave theory.
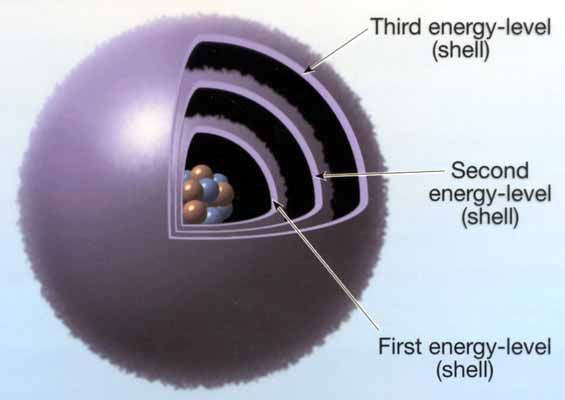
In the early days it was often asked, How large is a quantum of light? One answer is obtained by examining a star image formed with the great 100-inch reflector at Mt. Wilson. The diffraction pattern shows that each emission from each atom must be filling the whole mirror. For if one atom illuminates one part only and another atom another part only, we ought to get the same effect by illuminating different parts of the mirror by different stars (since there is no particular virtue in using atoms from the same star) ; actually the diffraction pattern then obtained is not the same. The quantum must be large enough to cover a 100-inch mirror.
But if this same star-light without any artificial concentration falls on a film of potassium, electrons will fly out each with the whole energy of a quantum. This is not a trigger action releasing energy already stored in the atom, because the amount of energy is fixed by the nature of the light, not by the nature of the atom. A whole quantum of light energy must have gone into the atom and blasted away the electron. The quantum must be small enough to enter an atom.
I do not think there is much doubt as to the ultimate origin of this contradiction. We must not think about space and time in connection with an individual quantum; and the extension of a quantum in space has no real meaning. To apply these conceptions to a single quantum is like reading the Riot Act to one man. A single quantum has not travelled 50 billion miles from Sirius; it has not been 8 years on the way. But when enough quanta are gathered to form a quorum there will be found among them statistical properties which are the genesis of the 50 billion miles’ distance of Sirius and the 8 years’ journey of the light.
The contradiction about the size of light quantum comes about when we consider it in terms of material-space and material-time. According to Einstein’s papers on quantization and relativity, the space and time for light quanta are much less condensed than the material-space and material-time.
The classical laws are based on the material substance, material-space and material-time. Even when light is not material (it is physical), it was treated only in context of material substance.
As science went deeper into the properties of light and electromagnetic phenomena, it ran into the property of quantization. The electromagnetic spectrum revealed a new substance, which may be called “field-substance”. The field-substance acted as continuous wave at lower frequencies, but with increased frequency it became condensed and acted more like a particle. Ultimately, the field-substance condensed to form the material-substance as in the nucleus of the atom.
Classical mechanics did not have to deal with quantization because it did not deal with field-substance. The New Quantum Theory was then developed to deal with field-substance.
.
Wave-Theory of Matter
It is comparatively easy to realise what we have got to do. It is much more difficult to start to do it. Before we review the attempts in the last year or two to grapple with this problem we shall briefly consider a less drastic method of progress initiated by De Broglie. For the moment we shall be content to accept the mystery as a mystery. Light, we will say, is an entity with the wave property of spreading out to fill the largest object glass and with all the well-known properties of diffraction and interference; simultaneously it is an entity with the corpuscular or bullet property of expending its whole energy on one very small target. We can scarcely describe such an entity as a wave or as a particle; perhaps as a compromise we had better call it a “wavicle”.
We misunderstand light by defining its wavelength, period and cycles in material units. A light quantum is the energy per cycle in light-units.
There is nothing new under the sun, and this latest. volte-face almost brings us back to Newton’s theory of light—a curious mixture of corpuscular and wave-theory. There is perhaps a pleasing sentiment in this “return to Newton”. But to suppose that Newton’s scientific reputation is especially vindicated by De Broglie’s theory of light, is as absurd as to suppose that it is shattered by Einstein’s theory of gravitation. There was no phenomenon known to Newton which could not be amply covered by the wave-theory; and the clearing away of false evidence for a partly corpuscular theory, which influenced Newton, is as much a part of scientific progress as the bringing forward of the (possibly) true evidence, which influences us to-day. To imagine that Newton’s great scientific reputation is tossing up and down in these latter-day revolutions is to confuse science with omniscience.
The wave-particle confusion with respect to light is resolved by the property of quantization discovered by Einstein.
To return to the wavicle.—If that which we have commonly regarded as a wave partakes also of the nature of a particle, may not that which we have commonly regarded as a particle partake also of the nature of a wave? It was not until the present century that experiments were tried of a kind suitable to bring out the corpuscular aspect of the nature of light; perhaps experiments may still be possible which will bring out a wave aspect of the nature of an electron.
So, as a first step, instead of trying to clear up the mystery we try to extend it. Instead of explaining how anything can possess simultaneously the incongruous properties of wave and particle we seek to show experimentally that these properties are universally associated. There are no pure waves and no pure particles.
The discovery that there are no pure waves and no pure particles, as made by de Broglie, supports the fundamental perspective of “continuum of substance”. One special case of this broad perspective that applies only to material-substance is the “particles in void” perspective.
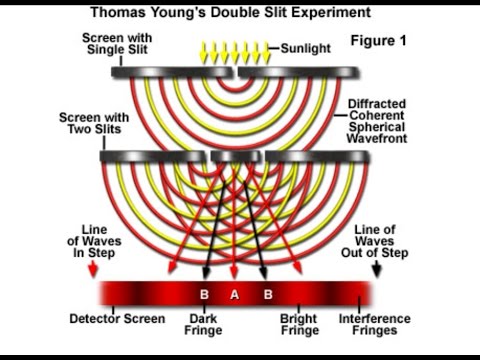
The characteristic of a wave-theory is the spreading of a ray of light after passing through a narrow aperture —a well-known phenomenon called diffraction. The scale of the phenomenon is proportional to the wavelength of the light. De Broglie has shown us how to calculate the lengths of the waves (if any) associated with an electron, i.e. considering it to be no longer a pure particle but a wavicle. It appears that in some circumstances the scale of the corresponding diffraction effects will not be too small for experimental detection. There are now a number of experimental results quoted as verifying this prediction. I scarcely know whether they are yet to be considered conclusive, but there does seem to be serious evidence that in the scattering of electrons by atoms phenomena occur which would not be produced according to the usual theory that electrons are purely corpuscular. These effects analogous to the diffraction and interference of light carry us into the stronghold of the wave-theory. Long ago such phenomena ruled out all purely corpuscular theories of light; perhaps to-day we are finding similar phenomena which will rule out all purely corpuscular theories of matter.*
*The evidence is much stronger now than when the lectures were delivered.
One cycle in light units shall appear as many cycles in material units. Using de Broglie’s method to calculate wavelengths from diffraction of waves, we may be able to find the ratio of light-units to material units for lengths. This shall reveal how much length shrinks from light frequency to material frequency.
A similar idea was entertained in a “new statistical mechanics” developed by Einstein and Bose—at least that seems to be the physical interpretation of the highly abstract mathematics of their theory. As so often happens the change from the classical mechanics, though far-reaching in principle, gave only insignificant corrections when applied to ordinary practical problems. Significant differences could only be expected in matter much denser than anything yet discovered or imagined. Strange to say, just about the time when it was realised that very dense matter might have strange properties different from those expected according to classical conceptions, very dense matter was found in the universe. Astronomical evidence seems to leave practically no doubt that in the so-called white dwarf stars the density of matter far transcends anything of which we have terrestrial experience; in the Companion of Sirius, for example, the density is about a ton to the cubic inch. This condition is explained by the fact that the high temperature and correspondingly intense agitation of the material breaks up (ionises) the outer electron systems of the atoms, so that the fragments can be packed much more closely together. At ordinary temperatures the minute nucleus of the atom is guarded by outposts of sentinel electrons which ward off other atoms from close approach even under the highest pressures; but at stellar temperatures the agitation is so great that the electrons leave their posts and run all over the place. Exceedingly tight packing then becomes possible under high enough pressure. R. H. Fowler has found that in the white dwarf stars the density is so great that classical methods are inadequate and the new statistical mechanics must be used. In particular he has in this way relieved an anxiety which had been felt as to their ultimate fate; under classical laws they seemed to be heading towards an intolerable situation—the star could not stop losing heat, but it would have insufficient energy to be able to cool down!**
** The energy is required because on cooling down the matter must regain a more normal density and this involves a great expansion of volume of the star. In the expansion work has to be done against the force of gravity.
The matter inside white dwarf stars is much denser than ordinary matter because it involves more dense packing of atomic nuclei. Such dense matter shall have higher quantization level compared to ordinary matter.
.