Reference: Subject Clearing Universe
NOTE: The subject of Physics treats these fundamental particles mathematically, and it does not provide a proper visualization for them. The effort on this blog is to provide a visualization that is consistent with what is known. It does not change the mathematics of Physics.
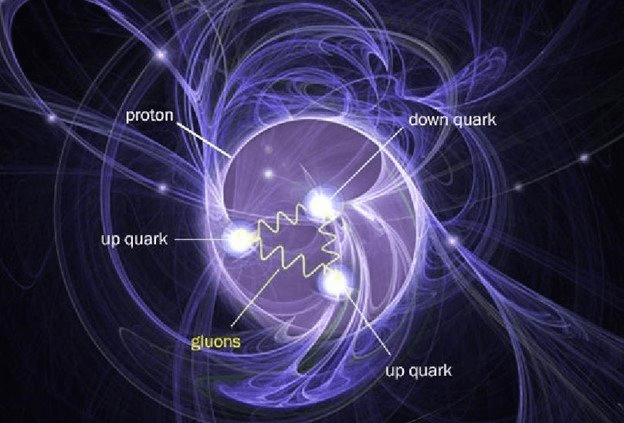
The charged proton layer exists at the surface of the nucleus. The subsequent layers in the interior of the nucleus are uncharged. They get more condensed as they get closer to the center of the nucleus.
.
Definition of NEUTRON
Beyond the outer proton layer, the inner layers of the nucleus are uncharged and more condensed. The neutrons that are ejected from these inner layers also have a precise vortex configuration. They have slightly more mass than the protons. The mass of a free neutron is measured as 1.675 × 10−27 kilograms. It has no charge.
Like the electron and the proton, the neutron does not exist as an independent particle within the atom because it is integrated with rest of the atom into layers of condensing substance. Therefore, it is an error to visualize neutrons as “particles” within the atom.
.
Misconception
1. It is a misconception to think that neutrons exist as independent “particles” within the atom. Neutrons form the layers very condensed substance in the interior of the nucleus.
Reference: Subject Clearing Universe
NOTE: The subject of Physics treats these fundamental particles mathematically, and it does not provide a proper visualization for them. The effort on this blog is to provide a visualization that is consistent with what is known. It does not change the mathematics of Physics.
Proton refers to that consistency of substance that is under the tension to evaporate from mass back into electronic liquidity. This tension is seen as positive charge.
.
Definition of PROTON
Protons form the surface layer of the nucleus. The charge of the proton comes from the fact that it is the substance that has condensed into mass from the electronic substance, and it can evaporate back into its previous electronic consistency. This proton layer is in equilibrium with the layer of the electronic substance immediately surrounding it.
The charge exists, because we have two very different consistencies in equilibrium, such that, the substance is being continuously exchanged between them. The exchanged substance appears as transitory particles listed under Leptons and Baryons.
When the proton layer separates itself from the atom it appears as its own little vortex called a proton. A proton has a precise configuration as well. It has the limiting consistency of mass, which is measured as 1.673 × 10−27 kilograms. This is 1836 times more condensed than the nearest electronic layer. It is considered to have a unit positive charge measured as 1.602 × 10−19 coulombs. This is equal and opposite to the charge of an electron.
The proton, too, does not exist as an independent particle within the atom because it is integrated with rest of the atom into layers of condensing substance. Therefore, it is an error to visualize protons as “particles” within the atom.
.
Misconception
1. It is a misconception to think that protons exist as independent “particles” within the atom. Protons form the first layer of mass in the nucleus that is still increasing in consistency as neutrons toward the center. Proton is the first layer of the nucleus that is formed after the last electronic layer condenses.
.
Reference: Subject Clearing Universe
NOTE: The subject of Physics treats these fundamental particles mathematically, and it does not provide a proper visualization for them. The effort on this blog is to provide a visualization that is consistent with what is known. It does not change the mathematics of Physics.
The free flowing radiant energy is called electromagnetic radiation because it is found to consist of electric and magnetic fields. It has a whole range of properties as expressed through an electromagnetic spectrum.
.
Definition of ELECTROMAGNETIC RADIATION
Electromagnetic radiation, such as, light, is free flowing energy that has not yet condensed into spinning vortices. The consistency of the radiation is represented by its assigned “frequency.”
The electromagnetic spectrum presents different ranges of frequencies of radiation. From lowest to highest, these ranges are called radio waves, microwaves, terahertz radiation, Infrared radiation, visible radiation, ultraviolet radiation, X-rays and Gamma rays. Gamma rays have the highest consistency. As this consistency increases the gamma rays are likely to coalesce into spinning vortices, such as, the electrons.
The configuration of the atom exists in equilibrium with the electromagnetic spectrum. The gamma rays are seen to be emitted by the nucleus, and X-rays from inner electrons. Other frequencies of the spectrum may be seen to be emitted from the upper electronic region of the atom.
An electromagnetic radiation has a certain frequency. Any effort to change that frequency activates a restoring force similar to the inertia of matter. In an electromagnetic field, force exists because different frequencies exist side-by-side. The higher is the gradient of frequencies, the higher is the force.
.
Misconceptions
1. It is not perceived in Physics that the electromagnetic radiation is a part of the spectrum of substance. At the upper end of the electromagnetic spectrum is the spectrum of the quantum particles and matter. Below the electromagnetic spectrum is the spectrum of thought substance in the form of space.
.
Reference: Subject Clearing Universe
NOTE: The subject of Physics treats these fundamental particles mathematically, and it does not provide a proper visualization for them. The effort on this blog is to provide a visualization that is consistent with what is known. It does not change the mathematics of Physics.
Electron refers to that consistency of substance that is under the tension to crystallize into the solidification of mass. This tension is seen as negative charge.
.
Definition of ELECTRON
Electrons are considered to be constituents of atoms. To understand electrons, one must understand the atom first.
The atom is a vortex of substance in which energy exists in equilibrium with mass. The free flowing energy starts to form into spinning vortices of energy. The spinning vortex consists of gradually condensing layers of electronic substance. At the center of these layers, the energy starts to precipitate into mass. Thus comes about the configuration of the atom.
The condensing layers of electronic substance are in equilibrium, and they have a precise configuration. They have been defined mathematically as orbitals. There are sub-layers within these layers, which are spotted through their atomic spectra.
When the electronic substance separates itself from the atom it appears as its own little vortex called an electron. An electron has a precise configuration as well. It has the limiting consistency of energy, which is measured in “mass” as 9.108 × 10−31 kilograms. It is considered to have a unit negative charge measured as 1.602 × 10−19 coulombs. The charge of the electron comes from the fact that it has a consistency that is just about to condense into mass.
The electron does not exist as an independent particle within the atom because it is integrated with rest of the atom into layers of condensing substance. Therefore, it is an error to visualize electrons as “particles” within the atom.
.
Misconceptions
1. It is a misconception to think that electrons exist as independent “particles” within the atom. Electrons do not have a center of mass to make them into a matter particle, which is represented by a point. As a spinning vortex, an electron is thousands of times larger than the nucleus in an atom, simply because it is much less condensed. Electrons basically surround the nucleus in the form of layers.
.
Reference: Subject Clearing Universe
The particle of thought has to be something very basic and precise. We identify it as a concept.
.
Definition of CONCEPT
The particle of thought is a concept that has a precise definition. It is formed by mentally combining all the characteristics or particulars of something. It is defined as an abstract idea.
A concept is understood to be a fundamental building block underlying principles, thoughts and beliefs. Concepts play an important role in all aspects of cognition. As such, concepts are studied within such disciplines as linguistics, psychology, and philosophy, and these disciplines are interested in the logical and psychological structure of concepts, and how they are put together to form thoughts and sentences. The study of concepts has served as an important flagship of an emerging interdisciplinary approach, cognitive science.
.
Misconceptions
1. Concepts cannot be arbitrary. They fit into the oneness of thought. By nature they are a unit in themselves.
2. Concepts cannot be fuzzy and unclear. They are a part of the laws that apply to thought. Concepts must be very precise by nature.
3. Any fuzzy and unclear concept is an anomaly.
.